Miach Orthopaedics wins expanded FDA nod for Bear implant
Miach Orthopaedics announced today that the FDA cleared an expanded indication for the company’s Bear implant for ACL tears.

Miach Orthopaedics announced today that the FDA cleared an expanded indication for the company’s Bear implant for ACL tears.

Lungpacer Medical announced today that it received FDA investigational device exemption (IDE) to begin a trial for its AeroNova system.

The findings provide new drug targets for stopping the infection’s spread.
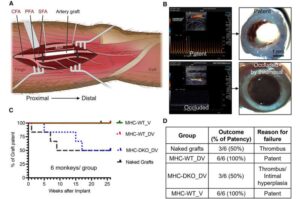
Scientists at the Wisconsin National Primate Research Center (WNPRC) and the Morgridge Institute for Research at the University of Wisconsin–Madison have been at the forefront of stem cell research and regenerative biology since James Thomson isolated the first human embryonic stem cell in 1998.

BREA, Calif., March 10, 2025 /PRNewswire/ — Beckman Coulter Diagnostics, a clinical diagnostics leader, today announced that the new DxC 500i Clinical Analyzer, an integrated clinical chemistry and immunoassay analyzer, received 510(k) clearance from the U.S. Food and Drug Administration. The DxC 500i combines advanced technology with an intuitive user interface, ensuring that laboratories of all sizes can meet the growing demands of modern healthcare. With throughput of up to 800 clinical chemistry tests per hour and 100 immunoassay tests per hour, this analyzer delivers precise and reliable results critical for timely clinical decision-making.