
Johnson & Johnson MedTech wins FDA nod for enhanced robotic-assisted bronchoscopy
Johnson & Johnson MedTech (NYSE: JNJ)+
announced today that the FDA cleared its Monarch Quest technology for robotic-assisted bronchoscopy.

Johnson & Johnson MedTech (NYSE: JNJ)+
announced today that the FDA cleared its Monarch Quest technology for robotic-assisted bronchoscopy.
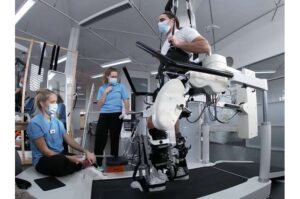
Spinal cord injuries are life-altering, often leaving individuals with severe mobility impairments. While rehabilitation robotics—devices that guide movement during therapy—have improved training for those with spinal cord injuries, their effectiveness remains limited. Without active muscle engagement, robotic-assisted movement alone does not sufficiently retrain the nervous system.

SALT LAKE CITY, March 12, 2025 /PRNewswire/ — For the thousands of patients in the United States requiring treatment for bone loss or defects caused by trauma or infections, Elute, Inc., a clinical stage company and emerging leader with a groundbreaking controlled and extended drug delivery platform, proudly announces U.S. Food and Drug Administration (FDA) approval of BonVie+™, a novel bone void filler implant.