
Avicenna.AI Launches AVI Seamless Platform for Medical Imaging AI
New platform is designed to enable fast, scalable access to a wide range of AI imaging solutions

New platform is designed to enable fast, scalable access to a wide range of AI imaging solutions

The clearance is based on data that included real-world evidence from 1,120 adolescents who received treatment at US TMS centres.

Renu MedTech today unveiled InsuPatch, its tubeless insulin patch pump designed specifically for advancing diabetes care in India.
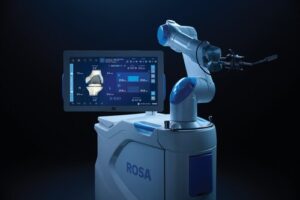
WARSAW, Ind., Nov. 14, 2025 /PRNewswire/ — Zimmer Biomet Holdings, Inc. (NYSE and SIX: ZBH), a global medical technology leader, today announced U.S. Food and Drug Administration (FDA) 510(k) clearance of ROSA® Knee with OptimiZe™, an enhanced version of its ROSA® Knee System that offers a more customized experience for surgeons to help deliver accurate and reproducible outcomes1 in robotic-assisted total knee replacement surgery.

Scientists at The Hospital for Sick Children (SickKids) have revealed a previously overlooked layer of genetic variation that could help explain why people experience disease differently, and why some treatments work better for certain populations.