QT Imaging secures FDA clearance for updated Breast Acoustic CT scanner
The scanner reconstructs 3D tomographic images by utilising both reflection-mode and transmission-mode ultrasound data.

The scanner reconstructs 3D tomographic images by utilising both reflection-mode and transmission-mode ultrasound data.
The dressing combines several technologies to minimise leakage, reduce dressing changes, and protect vulnerable skin.
Karl Storz announced that it received FDA clearance for the FIVE S 6.5 sterile single-use bronchoscope.
Now researchers in the former Laboratory of Chromatin Biology and Epigenetics, headed by C. David Allis until his death in early 2023, have discovered surprising new details about the mechanics of this protein complex, which could potentially lead to new cancer therapies. The findings were published in Genes & Development.

Researchers in the Soft Materials Laboratory (SMaL) in EPFL’s School of Engineering have now developed a way to 3D-print HA-based scaffolds using a room-temperature process that harnesses enzymes for fast mineralization.
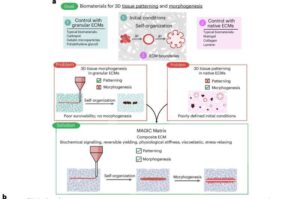
Miniature organs grown in the lab can organize themselves into complex shapes. But they never do it the same way twice, which makes it hard to use these so-called “organoids” to study disease. Now, scientists at UC San Francisco have created a new material that helps organoids grow in a more predictable way.

Now, a team of researchers from Brown University, Rhode Island Hospital, and VA Providence Healthcare has shown progress in restoring two-way communication across a damaged site of the spinal cord.
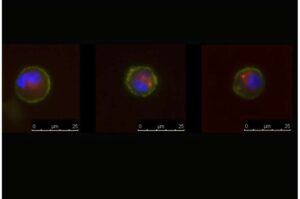
An enigmatic type of circulating tumor cell called a dual-positive (DP) cell is associated with shorter survival time in patients with advanced breast cancer, according to a study led by investigators at Weill Cornell Medicine and NewYork-Presbyterian.
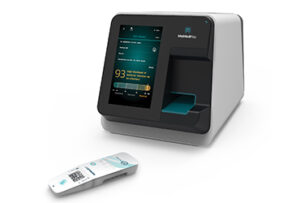
ANDOVER, Mass. and HAIFA, Israel, March 11, 2026 /PRNewswire/ — MeMed, a leader in advanced host-response diagnostics, today announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation (BDD) to MeMed BV Flex™, a test designed to accurately distinguish between bacterial and viral infections based on the body’s immune response.
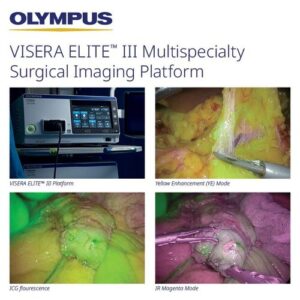
WESTBOROUGH, Mass., March 11, 2026 /PRNewswire/ — Olympus Corporation proudly announces the U.S. launch of the VISERA ELITE™ III surgical imaging platform designed to enhance surgical visualization, workflow efficiency, and multispecialty flexibility. The VISERA ELITE III platform is a significant leap forward in surgical imaging and the third major co-developed product by SONY Olympus Medical (SOMED), a joint venture established in 2013.