
JenaValve wins FDA nod for Trilogy heart valve system
JenaValve announced today that it received FDA premarket approval (PMA) for its Trilogy THV system.

JenaValve announced today that it received FDA premarket approval (PMA) for its Trilogy THV system.

Nia Therapeutics announced today that it received FDA breakthrough device designation for its Smart Neurostimulation System (SNS).

Dartmouth researchers have developed a cell phone-based tool that monitors tissue health by using a naturally occurring molecule to measure the oxygen level in cells.

A study has found that precise application of radio waves can change the activity of brain cells in ways that could counter neurological conditions.

Now, research published in ACS Central Science could help health care professionals non-invasively diagnose bacterial infections, using breath-based tests.
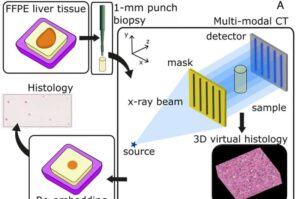
A new X-ray imaging technique could transform how hospitals analyze tissue samples, potentially speeding up diagnoses and improving outcomes for patients, shows a new study led by UCL researchers. The technology, developed in collaboration with the Memorial Sloan Kettering Cancer Center, Rigaku Americas and Creatv MicroTech, Inc., produces crisp 3D maps of biological tissue without cutting or staining samples, a significant improvement on the conventional process used in histopathology—the process of examining tissue to study, diagnose and treat diseases, particularly cancer.

Duke Health researchers show that a quick, outpatient nasal swab can pick up early biological changes linked to Alzheimer’s, even before thinking and memory problems appear.
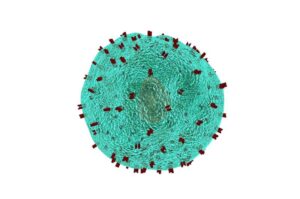
Now, scientists at UC San Francisco have developed a method to precisely reprogram these cancer-fighting cells directly inside the body, potentially eliminating the manufacturing process, cost, and waiting time that has kept this life-saving therapy out of reach for many patients around the world.

SUZHOU, China, March 18, 2026 /PRNewswire/ — Peijia Medical (9996.HK), a leading Chinese domestic company in the high-growth transcatheter valve therapeutics and neurovascular interventions markets, announced that its DCwire® Micro Guidewire has received 510(k) clearance from the U.S. Food and Drug Administration (“FDA”) on March 17, 2026. The product was independently designed and developed by Achieva Medical Limited, a subsidiary of the Company.
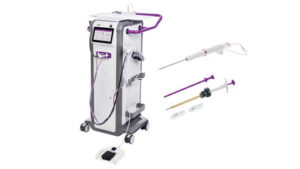
CINCINNATI, March 18, 2026 /PRNewswire/ — Mammotome, a Danaher company, announces FDA Clearance of two innovations for MR-guided breast biopsy: the Mammotome Prima™ MR Dual Vacuum-Assisted Breast Biopsy System – the industry’s first solution designed to be positioned in the MRI scanner room at the patient’s side – and the HydroMARK™ Plus Breast Biopsy Site Marker for MR, engineered for exclusive use with the Mammotome Prima™ system universal targeting set. Following a successful debut of the Mammotome Prima™ MR system in Europe, both products will be introduced to U.S. clinicians at the 2026 Society of Breast Imaging (SBI) Symposium. By bringing the device directly into the scanner room, the Mammotome Prima™ MR system enhances clinician coordination, streamlines communication, and helps maintain consistent focus on the patient throughout the procedure.