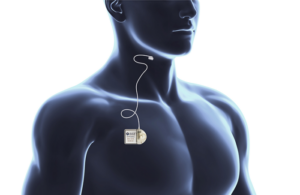
LivaNova wins FDA premarket approval for neuromod for sleep apnea
LivaNova (Nasdaq: LIVN)+ announced today that it received FDA premarket approval (PMA) for its aura6000 neuromodulation system for sleep apnea.

LivaNova (Nasdaq: LIVN)+ announced today that it received FDA premarket approval (PMA) for its aura6000 neuromodulation system for sleep apnea.
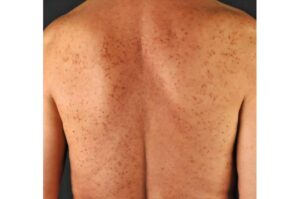
There is a promising new drug for the rare disease mastocytosis, which is associated with skin lesions, among other things. Researchers at the University of Basel have now been able to use artificial intelligence to quantitatively measure for the first time the extent to which it reduces skin lesions.

Researchers at Graz University of Technology (TU Graz) have developed a novel virtual reality (VR) system that could make the treatment of arachnophobia, also known as spider phobia, more targeted and personalized in the future. The work is published in the journal Frontiers in Human Neuroscience.