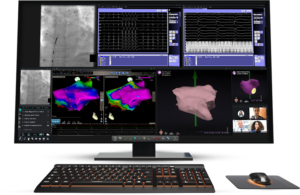
Stereotaxis wins FDA clearance for Synchrony cath lab tech
Stereotaxis (NYSE:STXS) announced today that it received FDA 510(k) clearance for its Synchrony system.

Stereotaxis (NYSE:STXS) announced today that it received FDA 510(k) clearance for its Synchrony system.

With approval to study the transcatheter replacement device in patients with severe tricuspid regurgitation, VDyne is advancing a treatment to compete with Edwards Lifesciences’ Evoque system.
Vantage’s intuitive three-step workflow, locate, dilate, decompress, enables physicians to perform procedures with consistency.
UCLA scientists have developed a simple and cost-effective blood test that, in early studies, shows promise in detecting multiple cancers, various liver conditions and organ abnormalities simultaneously by analyzing DNA fragments circulating in the bloodstream.