MedCAD’s AccuStride® Receives FDA 510(k) Clearance for New Anatomical Lower Leg Region Tib/Fib Solution
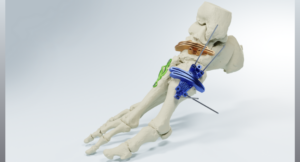
DALLAS, April 8, 2026 /PRNewswire/ — Dallas-based MedCAD has received FDA 510(k) clearance of its AccuStride® lower leg tibia and fibula system, making these patient-specific precision devices available to surgeons. The unique design of the custom-made planning and surgical guides will enable orthopedic surgeons to correct multiple related pathologies and osteotomies in a single procedure.