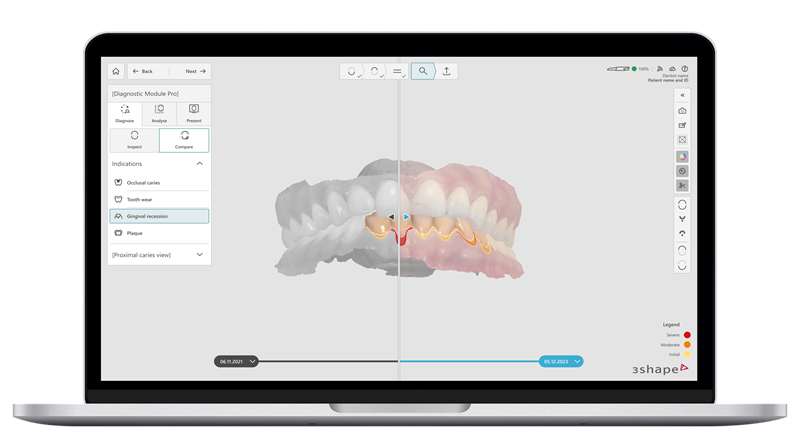
The 510(k) Premarket Notification allows Dx (R1) software to be commercially available in the United States for use by qualified dental professionals in dental clinics to aid in the diagnosis of key oral health conditions and the assessment of changes in teeth and gingiva in adult patients.
“I’m excited that we can now bring this transformative dental solution to doctors and patients across the United States,” says 3Shape CEO Jacob Paulsen. “Receiving FDA 510(k) clearance for Dx underscores our commitment to responsible innovation and to delivering digital solutions that meet the highest clinical and regulatory standards. With Dx software, dental professionals gain a powerful new way to communicate more clearly with patients about their oral health and treatment needs.”