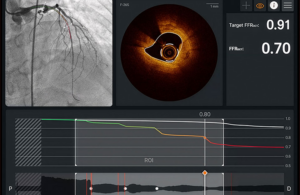
Ultreon 3.0 is an AI-powered imaging platform that uses optical coherence tomography (OCT). It combines the technologies in one system to better support patient outcomes and reduce risks in coronary imaging.
The combination of technologies gives doctors real-time planning guidance while they perform percutaneous coronary interventions (PCIs). It allows physicians to use OCT images to see the detailed structure of the blockage. By incorporating AI, they can assess the type of plaque causing the blockage. Altogether, this helps doctors choose the best location and stent size to improve blood flow, delivering faster, more precise procedures.