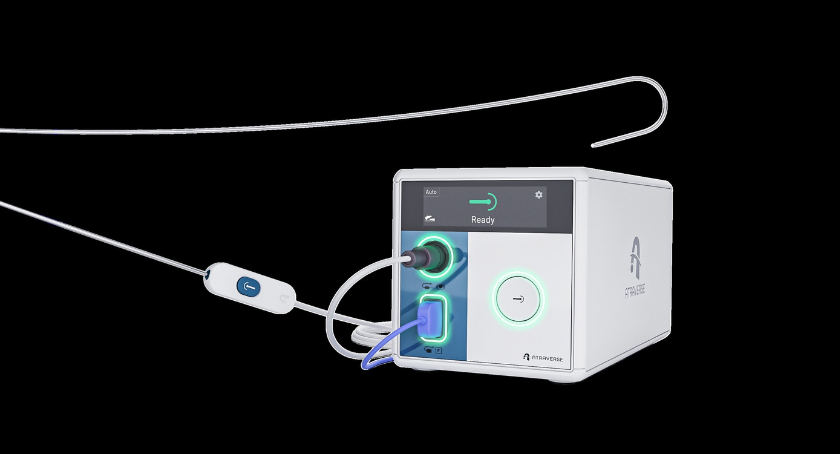
FULCRUM-VT evaluates the company’s vCLAS cryoablation technology for treating cardiovascular disease. Laguna Hills, California-based Adagio built its vCLAS cryoablation catheter on its proprietary ultra-low cryoablation technology (ULCT). It aims to address the challenges of ventricular tachycardia (VT) ablations, comprising part of the Adagio VT cryoablation system. The system’s features make it time- and effort-efficient across the range of purely endocardial ablation strategies in patients with multiple VT etiologies.
vCLAS picked up FDA breakthrough device designation in April 2025 and said a few months later that it completed enrollment for FULCRUM-VT.