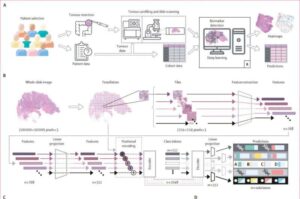
AI model simultaneously detects multiple genetic colorectal cancer markers in tissue samples
A multicenter study has analyzed nearly 2,000 digitized tissue slides from colon cancer patients across seven independent cohorts in Europe and the US. The samples included both whole-slide images of tissue samples and clinical, demographic, and lifestyle data.