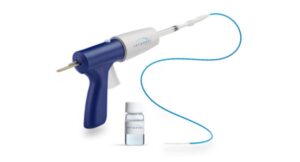
Teleflex wins FDA nod for expanded hemostatic device indications
Teleflex (NYSE: TFX) announced today that it received FDA 510(k) clearance for expanded indications of its QuikClot Control+ hemostatic device.

Teleflex (NYSE: TFX) announced today that it received FDA 510(k) clearance for expanded indications of its QuikClot Control+ hemostatic device.
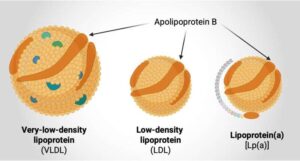
In a new study, led by Chalmers University of Technology in Sweden and Harvard University in the U.S., researchers have shown comprehensively that a combination of two lipoprotein markers, measured in a simple blood test, can give more accurate information about individual risk of heart disease than the current blood cholesterol test, potentially saving lives.

Urinary stents and catheters are implanted medical tubes that are widely used in human and veterinary medicine to drain urine to/from the bladder. Ureteral stents are used when the ureter, the duct between the kidney and bladder, is blocked by tumors, pregnancy, stones or anatomical narrowing.

Researchers from the University of Adelaide have developed a new technology for drug and functional genomics screenings, which could reshape the way diseases are treated.

Just one look at the next-generation lightweight, soft exoskeleton for children with cerebral palsy reveals the powerful role technology can play in solving global challenges and improving lives.
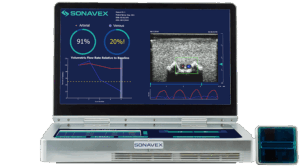
Sonavex announced that it received FDA 510(k) clearance for its AI-enabled EchoMap device with 3D ultrasound capabilities.
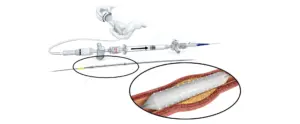
Orchestra BioMed (Nasdaq:OBIO) announced today that it received FDA investigational device exemption (IDE) for its Virtue drug-coated balloon.

A judge has issued a permanent injunction, signaling an Insulet (Nasdaq:PODD) victory in a trade secret case against EOFlow.
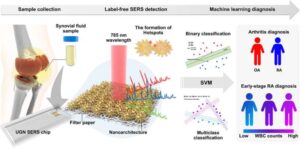
Dr. Ho Sang Jung and his research team from the Advanced Bio and Healthcare Materials Research Division at the Korea Institute of Materials Science (KIMS), in collaboration with Seoul St. Mary’s Hospital, have developed a technology that enables the diagnosis of osteoarthritis and rheumatoid arthritis within 10 minutes using synovial fluid. This marks the first such achievement in Korea.
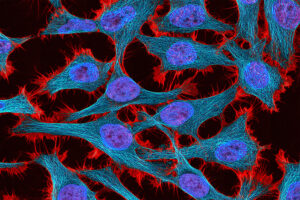
The circuits could help researchers develop new treatments for fragile X syndrome and other diseases caused by mutations of a single gene.