
Edwards receives CE mark for Sapien M3 mitral valve replacement device
The replacement system is launching into markets already served by repair devices, namely Edwards’ Pascal and Abbott’s Mitraclip.

The replacement system is launching into markets already served by repair devices, namely Edwards’ Pascal and Abbott’s Mitraclip.
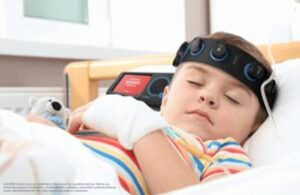
CeriBell (Nasdaq:CBLL) announced today that it received FDA 510(k) clearance for its next-generation Ceribell Clarity algorithm.
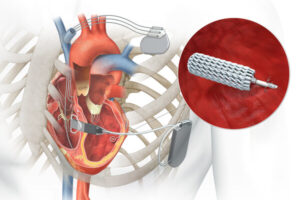
EBR Systems announced that it received FDA approval for its wireless cardiac pacing device for heart failure

DESKi today announced FDA clearance of HeartFocus, its AI-enabled heart exam software.
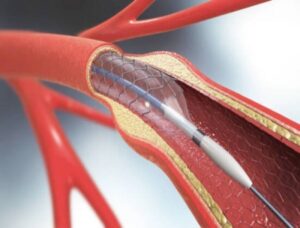
First Dual-Layer Micromesh Carotid Stent Approved for Use in the U.S.
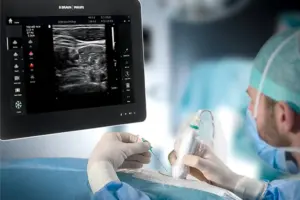
BETHLEHEM, Pa., April 14, 2025 /PRNewswire/ — B. Braun Medical Inc. (B. Braun), a leader in smart infusion therapy and pain management, announced today the launch of the EZCOVER® Probe Cover Set into the U.S. market.
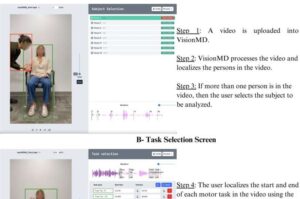
A University of Florida researcher has developed an open-source computer program that uses artificial intelligence to analyze videos of patients with Parkinson’s disease and other movement disorders. The tool, called VisionMD, helps doctors more accurately monitor subtle motor changes, improving patient care and advancing clinical research.
Even though the virus was first discovered in laboratory monkeys in 1958, the original source came from a squirrel.
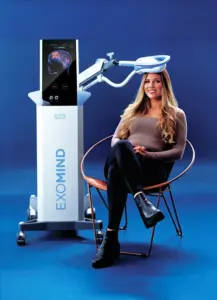
Non-Invasive TMS Device Aims to Enhance Mental Health by Targeting Neural Activity with Magnetic Pulses

Dexcom (Nasdaq: DXCM) today announced a significant regulatory milestone for its continuous glucose monitoring (CGM) technology.