
Nevro wins CE mark for HFX iQ spinal cord stim for pain relief
Nevro (NYSE:NVRO) announced today that it received CE mark in Europe for its HFX iQ spinal cord stimulation (SCS) system.

Nevro (NYSE:NVRO) announced today that it received CE mark in Europe for its HFX iQ spinal cord stimulation (SCS) system.
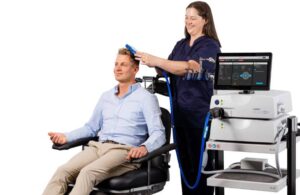
Magstim announced today that the FDA cleared its next-generation Horizon Inspire system for delivering transcranial magnetic stimulation (TMS).
iCad (Nasdaq:ICAD) announced today that the FDA cleared its ProFound Detection version 4.0 for digital breast tomosynthesis (DBT).
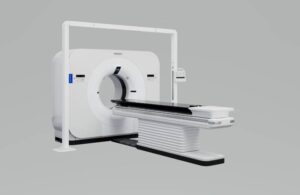
Philips (NYSE:PHG) announced today that the FDA granted 510(k) clearance for its new detector-based spectral computed tomography (CT) radiotherapy solution.

Fibrin sheath formation is a challenge for physicians using CVCs, as it blocks blood flow and makes CVCs less effective; the new AstutePlus prevents fibrin sheath formation.

MedTech startup Accunea has developed RenoSure, a new device that has been designed to ‘transform’ kidney care according to the company, and increase the number of organs available for transplantation.

MediKraft SealBase is a surface-treated medical paper that the company says is designed to meet the growing needs of the medical packaging industry.

TEL AVIV, Israel and FORT LAUDERDALE, Fla., Nov. 11, 2024 /PRNewswire/ — Momentis Surgical Ltd., a leader in robotic-assisted surgical innovation, today announced the U.S. Food and Drug Administration (FDA) 510(k) clearance for its second-generation Anovo® robotic surgical platform.

Avanos Medical announced today that a Centers for Medicare and Medicaid Services (CMS) payment rule benefits its infusion systems.

RNA in the human genome’s understudied ‘Wild West’ might be key to regulating genetic disorders like epilepsy, autism