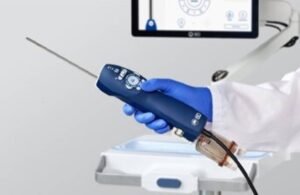
BD wins FDA nod for breast biopsy, tissue removal system
BD (NYSE: BDX)+
announced today that the FDA granted 510(k) clearance for its EnCore EnCompass breast biopsy and tissue removal system.

BD (NYSE: BDX)+
announced today that the FDA granted 510(k) clearance for its EnCore EnCompass breast biopsy and tissue removal system.
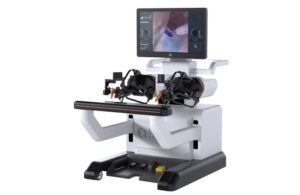
EndoQuest Robotics announced today that the FDA approved the initiation of the next and final stage of its PARADIGM trial.

Scientists have identified a promising target for treatment of a devastating autoimmune disease affecting the brain.
A new Brazilian study has revealed the key role of the protein periostin and stellate pancreatic cells in allowing pancreatic cancer to infiltrate nerves and spread early, increasing the risk of metastasis.
Diabetes, a metabolic disease, is on the rise worldwide, and over 90% of cases are type 2 diabetes, where the body does not effectively respond to insulin.
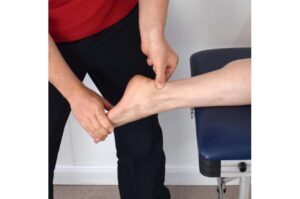
Complaints such as pain in the Achilles tendon, tennis elbow, swimmer’s shoulder and jumper’s knee are familiar to many young sportspeople, as well as to older individuals. These conditions are all caused by overloading of tendons and are generally very painful.
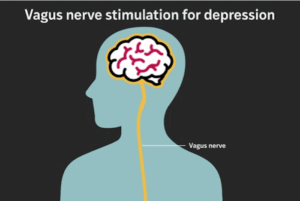
About 20% of U.S. adults experience major depression in their lifetime. For most people, symptoms improve within a few treatment attempts, but up to one‐third of patients have treatment‐resistant depression, for which standard antidepressant medication or psychotherapy isn’t enough.

The human brain is complex. Understanding deep brain function usually requires the insertion of probes that frequently result in irreversible tissue damage. Current neural probes are made out of silicon, a brittle material that can shatter during placement.
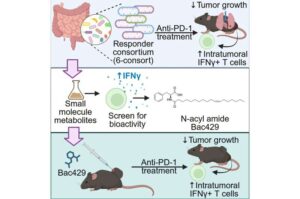
UF Health Cancer Institute researchers have discovered a small compound produced naturally by gut bacteria that doubled the response to lung cancer immunotherapy treatment in mice and can now be made into a drug for testing in humans.
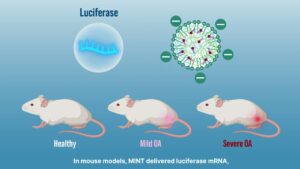
Nanoparticles are engineered to home in on areas where cartilage has degenerated in osteoarthritis, ensuring that treatment concentrates exactly where it is needed