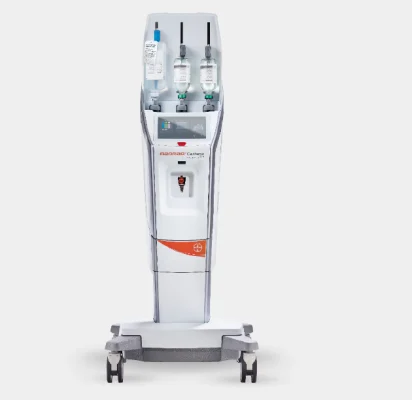
The US Food and Drug Administration (FDA) has granted expanded clearance to Bayer’s Medrad MRXperion, broadening the magnetic resonance (MR) injection system’s application across a wider range of MRI scanners.
Used for delivering contrast agents and saline during MRI diagnostic procedures, the FDA clearance expansion updates the labelled field strength compatibility of Medrad MRXperion to support MRI scanners with magnetic fields up to and including 7T (Tesla) range.
The FDA clearance also greenlights the injection system’s compatibility with Bayer’s Imaging Scanner Interface 2 (ISI2). The German company anticipates that the introduction of compatibility with I2I2, a hardware-software communication module for synchronising communication between the injection system and MRI scanner, will streamline MR exam coordination and drive operational efficiency in the MRI suite.