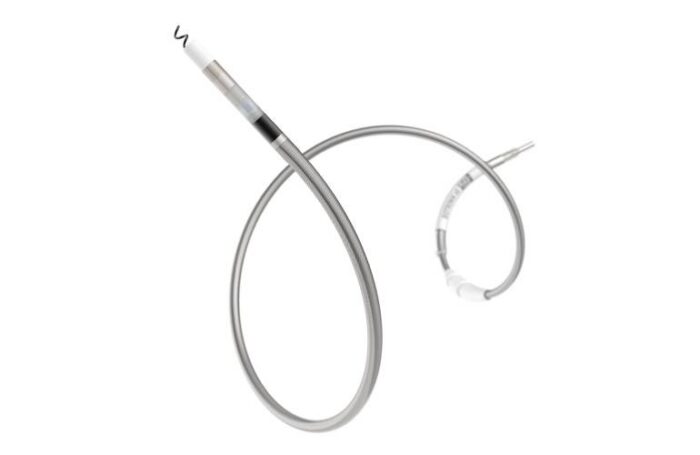
The company describes Solia CSP S as the world’s first pacing lead to combine the support of a style-driven lead platform with the efficiency of a fixed helix. According to Biotrinik, it’s the first pacing lead specifically designed for LBBAP.
Approval enables Biotronik to bring the system to the U.S. market as it seeks to compete against pacing leaders Abbott and Medtronic, which both delivered positive trial outcomes for their LBBAP systems last year.