MedTech News
.................... by Andrew Celentano

Medtronic begins full commercial rollout of MiniMed 780G with Instinct sensor made by Abbott
Medtronic (NYSE: MDT)+
today announced the full U.S. launch for its Instinct sensor, made by Abbott, paired with the MiniMed 780G pump.

FDA accepts MannKind sNDA for autoinjector that treats edema
MannKind (Nasdaq:MNKD) announced that the FDA accepted a supplemental New Drug Application (sNDA) for its Furoscix ReadyFlow autoinjector.

Cleveland Diagnostics’ IsoPSA secures FDA approval for prostate cancer
The decision is supported by evidence from a clinical study and data from supporting analytical validation trials.

Neusoft Medical Systems Unveils World’s First 8cm Wide-Coverage Photon-Counting CT
Neusoft Medical’s latest high-end systems are gaining rapid global traction, with installations rising worldwide
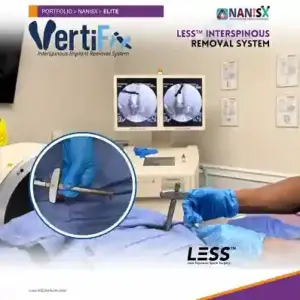
KIC Ventures Unveils VertiFix to Solve Growing Crisis in Interspinous Implant Revisions
KIC Ventures began educating physicians to use the InSpan instrumentation system to carefully dissect around interspinous implants and remove them with minimal disruption

Overlooked hormone may be deadly driver of postmenopausal breast cancer in women with obesity
A new analysis of research into the most common type of breast cancer has zeroed in on an overlooked hormone that may be responsible for the increased risk of breast cancer death in postmenopausal women with obesity.
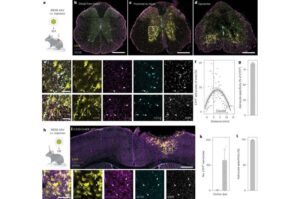
How the nervous system activates repair after spinal cord injury
After a spinal cord injury, cells in the brain and spinal cord change to cope with stress and repair tissue. A new study from Karolinska Institutet, published in Nature Neuroscience, shows that this response is controlled by specific DNA sequences. This knowledge could help develop more targeted treatments.

In-home sensor technology offers smarter care for ALS patients
Bill Janes is on a mission to improve life for people with amyotrophic lateral sclerosis (ALS). As a licensed occupational therapist and researcher at the University of Missouri, he’s seen firsthand how the disease can steal a person’s strength, speech and independence.
