MedTech News
.................... by Andrew Celentano
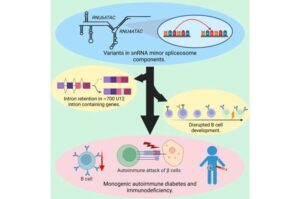
Overlooked non-coding genes cause diabetes in babies, study reveals
Scientists have found new genetic causes for diabetes in babies—in a part of the genome that has historically been overlooked in genetic studies.
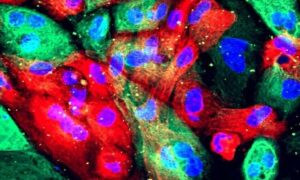
Long non-coding RNA may be a promising therapeutic target for cancer
Northwestern Medicine scientists have discovered that a specific long non-coding RNA activates oncogenic signaling pathways in prostate cancer cells and drives tumor progression, underscoring its potential as a therapeutic target, according to a recent study published in Nature Communications.
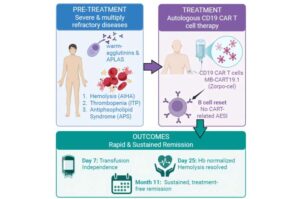
CAR-T therapy drives remission in patient with three autoimmune diseases
For the first time, scientists have used a modern cell therapy called CAR-T to treat a patient with three different life-threatening autoimmune diseases that had resisted years of treatment.

Sauna heat sends white blood cells surging through your bloodstream, study finds
Sauna bathing releases white blood cells into the bloodstream, a new study from Finland shows. Circulating white blood cells play a key role in the body’s defense against various pathogens and diseases. The results were published in the journal Temperature.
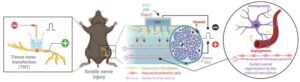
Novel gene-based therapy helps nerves heal better after severe injury
“This study is the first to combine tissue nanotransfection (TNT) with nerve graft surgery, and the results are promising.
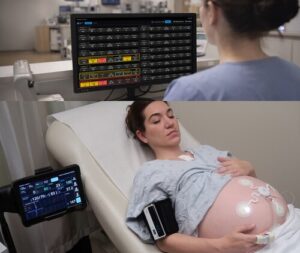
Sibel Health Receives FDA-Clearance for ANNE® Maternal, a Comprehensive and Fully Wireless Maternal-Fetal Monitoring Platform
Sibel Health today announced FDA 510(k) clearance of ANNE® Maternal, the world’s first fully wireless, comprehensive maternal-fetal monitoring platform.

InventHelp Inventors Develop New Ovulation Monitoring Device (RKH-979)
PITTSBURGH, April 8, 2026 /PRNewswire/ — “Our intention is to create a more accurate and exact wearable device to determine the onset of the female ovulation cycle and consequently when a female human or animal is most likely to become pregnant after insemination,” stated one of the two inventors, from Gainesville, Virginia, “hence the invention of the Ovulonics device. Our design would provide an easy, convenient and accurate detection time for human or animal
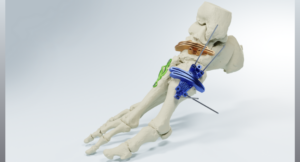
MedCAD’s AccuStride® Receives FDA 510(k) Clearance for New Anatomical Lower Leg Region Tib/Fib Solution
DALLAS, April 8, 2026 /PRNewswire/ — Dallas-based MedCAD has received FDA 510(k) clearance of its AccuStride® lower leg tibia and fibula system, making these patient-specific precision devices available to surgeons. The unique design of the custom-made planning and surgical guides will enable orthopedic surgeons to correct multiple related pathologies and osteotomies in a single procedure.
