MedTech News
.................... by Andrew Celentano
Glucotrack picks up new patents for continuous blood glucose monitor
Glucotrack (Nasdaq:GCTK) announced today that it received new U.S. patents for its continuous blood glucose monitoring (CBGM) platform.

Top medtech trends to watch in 2026
From M&A to surgical robotics and user fee negotiations, the medical device industry has a busy year ahead. Check out MedTech Dive’s roundup of the top medtech trends to watch in 2026.
RevBio receives FDA clearance for cranial bone glue trial
The FDA has authorised up to 15 clinical sites to take part in the study.

Imperative Care launches new Zoom 4S catheter
Imperative Care announced the launch of the new Zoom 4S thrombectomy catheter and reported the first patient cases with the device.

The top 10 cardio device stories of 2025
Pulsed-field ablation, transcatheter heart valves, and intravascular lithotripsy are among the top stories, as they drive growth among the largest cardio device companies.
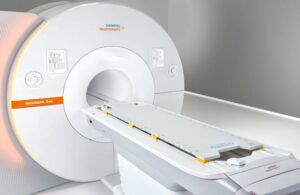
Siemens Healthineers wins FDA nod for PET/MR imaging scanner
Siemens Healthineers announced today that it received FDA clearance for its Biograph One second-generation PET/MR scanner.

Senseonics wins CE mark for Eversense 365 year-long CGM
Senseonics (NYSE:SENS) announced today that its Eversense 365 continuous glucose monitor (CGM) system received CE mark approval.
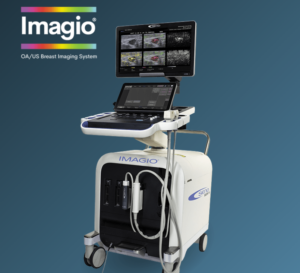
Seno Medical’s Next-Generation Imagio® Imaging System Obtains European Union (EU) Medical Device Regulation (MDR) CE Mark Certification
The CE Mark certification enables Seno Medical to market and sell the latest version of its Imagio® Imaging System with opto-acoustic, sound, and artificial intelligence in the European Union.
