MedTech News
.................... by Andrew Celentano
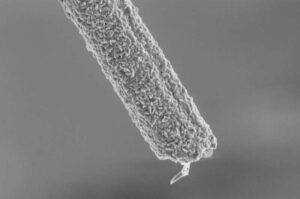
New diamond-coated electrodes may help people walk again
What’s the first thing you did when you woke up this morning? Maybe you swung your legs over the side of your bed, placed your feet on the floor and stood up. Simple, right?
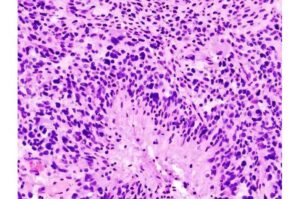
Scientists uncover hidden cells fueling brain cancer—and a drug that could stop them
A team of Canadian scientists has uncovered a new way to slow the growth of glioblastoma, the most aggressive and currently incurable form of brain cancer—and identified an existing medication that could treat it.
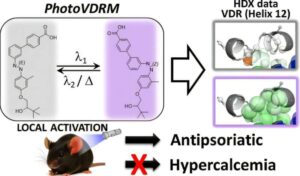
Treating psoriasis without side effects: Light-activated molecules demonstrate potential
Researchers from the Institute for Advanced Chemistry of Catalonia (IQAC) of the Spanish National Research Council (CSIC) have developed a series of light-activatable drug candidates that could serve as a potential treatment for psoriasis.
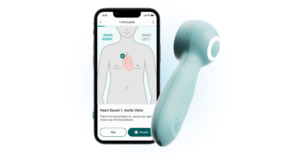
Covestro delivers new solution for in-home electronic stethoscope
The project began at the University of Pittsburgh’s Makerspace during the Pitt Challenge Healthcare Hackathon in 2021 and has since grown into a community driven effort.

Study reveals potential therapeutic target for treating glaucoma
Researchers have identified a metabolically sensitive cell subtype in the eye’s drainage system which shows early signs of dysfunction in a genetic mouse model of glaucoma.
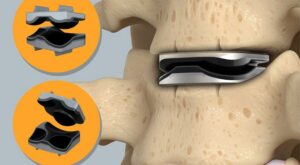
Omnia Medical Announces Commercial Launch of FDA-Cleared PsiF DNA™ System
MORGANTOWN, W.Va., Jan. 20, 2026 /PRNewswire/ — Omnia Medical, a medical technology company developing surgical solutions for spine and interventional pain physicians, today announced the commercial launch of its FDA-cleared PsiF DNA™ Sacroiliac Joint Stabilization System.
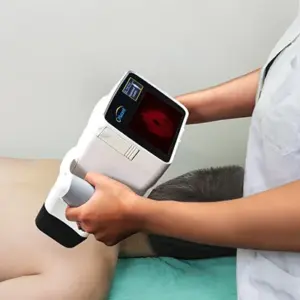
Clinical Trial Demonstrates Accurate Non-Invasive Detection of In Vivo Biologic Activity in Melanomas by the Orlucent® Skin Fluorescent Imaging System
LOS GATOS, Calif., Jan. 20, 2026 /PRNewswire/ — In the pivotal clinical trial of their technology, researchers at Orlucent®, Inc. have confirmed the accuracy of the company’s Skin Fluorescent Imaging (SFI) System, a non-invasive, hand-held molecular imaging device for the direct, on-the-skin, point-of-care assessment of suspicious moles and early detection of melanoma-related activity.

Cepheid Receives FDA Clearance for Xpert® GI Panel to Support Broad Detection of Gastrointestinal Pathogens
FDA-cleared PCR test aids in detection of 11 diarrhea-causing bacteria, viruses, and parasites from 1 sample
