MedTech News
.................... by Andrew Celentano
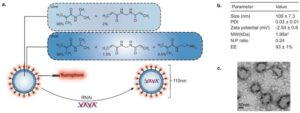
Nanohydrogels steer cancer drugs to tumors, aiming to spare healthy tissue
Cancer cells consume oxygen and nutrients at a higher rate than healthy tissue,. This drug delivery system senses those physical changes and guides medicine to the disease. The drug is released only when it encounters those tumor-specific conditions.
Brain immune cells may help build Alzheimer’s plaques
A new study led by researchers from VIB and KU Leuven shows that immune cells called microglia can actively promote the formation of plaques in Alzheimer’s disease.
Study identifies gene linked to chemotherapy resistance in prostate cancer
A gene called FOXJ1 may drive resistance to taxane chemotherapy during treatment for advanced prostate cancer, according to a new study led by investigators at Weill Cornell Medicine and Beth Israel Deaconess Medical Center.

Real-time metabolic monitoring on a chip: What happens inside a cell can be measured instantly
In a significant advancement for lab-on-chip technology, IBEC researchers in the frame of the European project BLOC, have demonstrated the first integration of a benchtop nuclear magnetic resonance (NMR) spectrometer with a microfluidic cell culture platform capable of real-time metabolic monitoring,
Blood test predicts dementia in women as many as 25 years before symptoms begin
Researchers from the University of California San Diego have found that a novel blood-based biomarker can predict a woman’s risk of developing dementia as many as 25 years before symptoms appear.
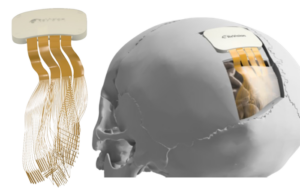
ReVision Implant wins FDA breakthrough nod for vision-restoring BCI
ReVision Implant announced today that it received FDA breakthrough device designation for its Occular visual cortical prosthesis.
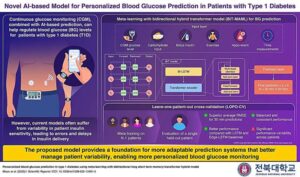
AI model enables personalized blood glucose predictions for type one diabetes
A research team led by Professor Jaehyuk Cho from the Department of Software Engineering at Jeonbuk National University in South Korea, have developed an innovative model, named BiT-MAML, aimed at tackling inter-patient variability in BG prediction.

New 3D material enables brain cells to communicate
Researchers at Kiel University (CAU) have now developed a novel 3D material on which human brain cells can grow and exchange signals in the lab.
