
FDA grants speedy approval to Eli Lilly’s weight-loss pill for obesity
Federal regulators on Wednesday approved Eli Lilly’s new weight-loss pill, a second daily oral medication to treat obesity and other weight-related conditions.

Federal regulators on Wednesday approved Eli Lilly’s new weight-loss pill, a second daily oral medication to treat obesity and other weight-related conditions.
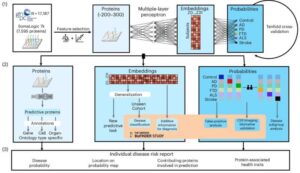
“Our hope is to be able to accurately diagnose several diseases at once with a single blood test in the future,” says Vogel, who led the study. He is an assistant professor, head of a research group, and part of the strategic research area MultiPark at Lund University.

Garmin users can now integrate with the temperature-powered Natural Cycles app, which turns Garmin skin temperature data into fertility insights for women’s health
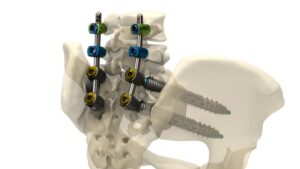
AUSTIN, Texas, March 31, 2026 /PRNewswire/ — Genesys Spine today announced the successful completion of the first surgical procedure utilizing the SIros-X System, a next-generation solution designed to deliver precision, adaptability, and control for complex sacroiliac joint fusion and pelvic fixation. The inaugural case was performed by Scott L. Parker, M.D., at Ascension Saint Thomas Hospital Midtown, Nashville, TN, marking an important milestone as the system begins clinical use.
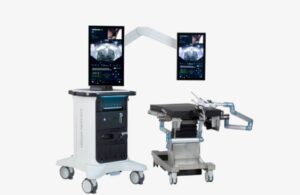
Procept BioRobotics (Nasdaq:PRCT) today announced the international rollout of its next-generation Hydros robotic system for Aquablation therapy.
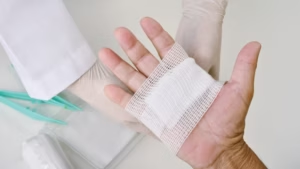
The dressing combines several technologies to minimise leakage, reduce dressing changes, and protect vulnerable skin.
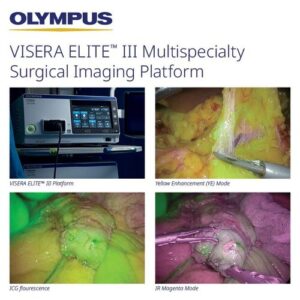
WESTBOROUGH, Mass., March 11, 2026 /PRNewswire/ — Olympus Corporation proudly announces the U.S. launch of the VISERA ELITE™ III surgical imaging platform designed to enhance surgical visualization, workflow efficiency, and multispecialty flexibility. The VISERA ELITE III platform is a significant leap forward in surgical imaging and the third major co-developed product by SONY Olympus Medical (SOMED), a joint venture established in 2013.

MiniMed (Nasdaq:MMED) announced that it received CE mark for the use of its MiniMed 780G with the Instinct sensor made by Abbott.
New product line extends options to better meet patient needs
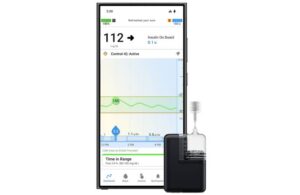
Tandem Diabetes Care (Nasdaq:TNDM) announced today that it made its Mobi automated insulin delivery system available with Android devices.