
Axogen announces first surgical implant of Avive+ Soft Tissue Matrix
Axogen (Nasdaq:AXGN) this week announced the first surgical implantation of its Avive+ Soft Tissue Matrix.

Axogen (Nasdaq:AXGN) this week announced the first surgical implantation of its Avive+ Soft Tissue Matrix.

Inspira announced that it has received the Canadian Standards Association (CSA) Certification of Compliance to U.S. standards for the INSPIRA ART100 device.
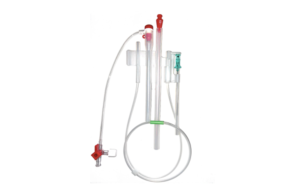
Front Line Medical Technologies today announced it received CE mark approval for its COBRA-OS device.
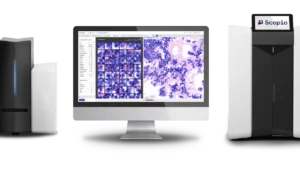
Scopio can now add the application to its imaging platforms that allow users to view blood samples digitally rather than under a microscope.

Highmark BCBS and BCBS of North Dakota cover the Leva Pelvic Health System for women seeking effective treatment for urinary incontinence
Vivos Therapeutics specializes in the development and commercialization of highly effective proprietary treatments for sleep related breathing disorders (including all severities of obstructive sleep apnea (OSA) in adults).

Medical Microinstruments said the system could increase the number of physicians who can perform complicated microsurgical procedures.

New Indication for Treatment of Pulmonary Embolism Enhances Device Utility in Critical Medical Scenarios
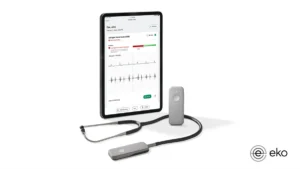
The clearance adds to the list of devices the FDA has authorized this year with AI algorithms to detect health conditions.

Biotronik this week announced it received FDA breakthrough device designation for its Freesolve below-the-knee resorbable magnesium scaffold (BTK RMS).