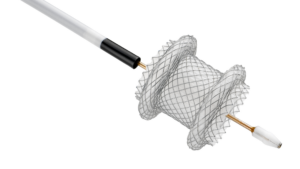
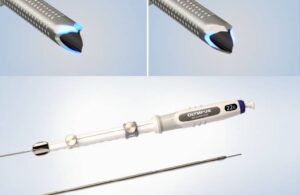
Vesalio Expands International Neurovascular Portfolio with CE Mark of NeVa VS and NeVa 3.0 mm and Receives Additional FDA 510(k) Clearance for its Aspiration Catheters
PLANO, Texas, Feb. 10, 2026 /PRNewswire/ — Vesalio, a global leader in vascular intervention, today announced CE Mark certification and the European commercial launch of two new neurovascular devices: NeVa™ VS, for the treatment of cerebral vasospasm following aneurysmal subarachnoid hemorrhage (aSAH), and the NeVa™ 3.0 mm Thrombectomy System for stroke. In addition, the Company received an additional U.S. Food and Drug Administration (FDA) 510(k) clearance expanding the indications of its neurovascular and peripheral aspiration catheters to include distal access.