FDA approves Anumana’s pulmonary hypertension algorithm
The algorithm integrates with EHR systems and ECG platforms, operating within the health system without external data transfer.

The algorithm integrates with EHR systems and ECG platforms, operating within the health system without external data transfer.
BOSTON, March 30, 2026 /PRNewswire/ — Haemonetics Corporation (NYSE: HAE), a global medical technology company focused on delivering innovative solutions designed to improve patient outcomes, today announced U.S. Food and Drug Administration (FDA) approval to expand the labeling for the VASCADE MVP® XL venous vascular closure system to include procedures using 10-14F inner diameter (ID) and up to 17F outer diameter (OD) procedural sheaths. With this label expansion, the VASCADE MVP XL system is approved for larger sheaths used in market-leading technologies for pulsed field ablation (PFA) and left atrial appendage closure (LAAC) to treat atrial fibrillation.

New technology designed to provide a single irrigation management system for ureteroscopy, cystoscopy, percutaneous nephrolithotomy (PCNL) and benign prostatic hyperplasia (BPH) procedures
4DMedical Limited (ASX:4DX), the global leader in cardiothoracic imaging software, announces that its latest imaging technology, CT:VQ™ has received CE Mark certification for commercial use in the European Union.
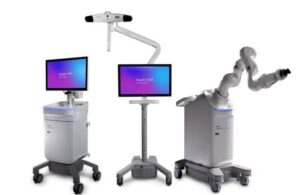
Medtronic (NYSE:MDT) announced today that the FDA cleared its Stealth AXiS surgical robotic system for new indications.

Novo Nordisk announced today that it received FDA approval for Awiqli, the first and only once-weekly, long-acting basal insulin.
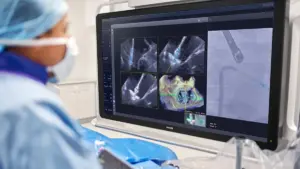
The system uses AI to optimize the placement of Edwards’ mitral valve repair device.

An Abbott (NYSE:ABT) official has shared a significant reimbursement win for its Esprit BTK drug-eluting scaffold system.

Noah Labs Vox uses AI algorithms to detect worsening heart failure based on patients’ voice recordings.

Achieving MDR compliance shows Modius Sleep meets safety, clinical, and quality standards required for entry into the EU market.