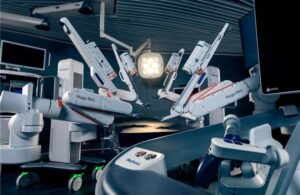
Medtronic gets FDA nod for Hugo surgical robot in urologic procedures
Medtronic (NYSE: MDT)+
announced today that the FDA cleared its Hugo robotic-assisted surgery system for use in urologic surgical procedures.

Medtronic (NYSE: MDT)+
announced today that the FDA cleared its Hugo robotic-assisted surgery system for use in urologic surgical procedures.
The test, which runs on the cobas liat system, is said to deliver results in 15 minutes.
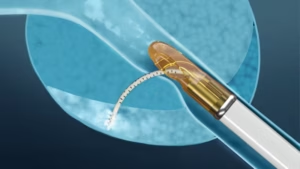
The Vanquish device’s clinical data demonstrated an elimination of MRI visible intermediate risk disease in 91% of patients after a single procedure.
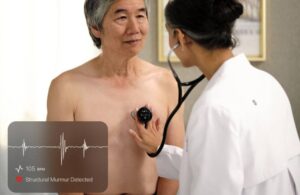
Eko Health announced today that the Centers for Medicare & Medicaid Services (CMS) has finalized national payment for its Sensora platform.

MannKind (Nasdaq:MNKD) announced that the FDA accepted a supplemental New Drug Application (sNDA) for its Furoscix ReadyFlow autoinjector.

The decision is supported by evidence from a clinical study and data from supporting analytical validation trials.

Medical Microinstruments (MMI) announced today that it won reimbursement for its surgical lymphovenous bypass (LVB) surgery system.
The a2z-Unified-Triage device is designed to simultaneously flag and prioritise seven urgent findings on abdomen-pelvis CT scans.

GE HealthCare’s Recon DL is claimed to be the first mammography technology to use deep learning alongside iterative reconstruction to improve image quality.

BD Surgiphor™ is a sterile, ready-to-use irrigation solution designed to help loosen and remove debris from surgical wounds during procedures