Siemens Healthineers wins FDA nod for PET/MR imaging scanner
Siemens Healthineers announced today that it received FDA clearance for its Biograph One second-generation PET/MR scanner.

Siemens Healthineers announced today that it received FDA clearance for its Biograph One second-generation PET/MR scanner.

Senseonics (NYSE:SENS) announced today that its Eversense 365 continuous glucose monitor (CGM) system received CE mark approval.
The CE Mark certification enables Seno Medical to market and sell the latest version of its Imagio® Imaging System with opto-acoustic, sound, and artificial intelligence in the European Union.

Glaukos (NYSE:GKOS) announced today that the FDA approved a new drug application (NDA) labeling supplement for its iDose TR system.
eMurmur has received FDA 510(k) clearance for its next-generation heart murmur detection software, eMurmur Heart AI (2.2).
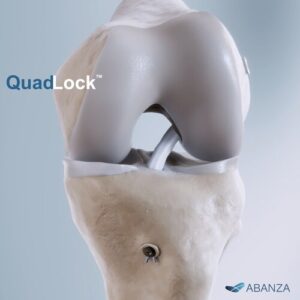
ORLANDO, Fla., Jan. 27, 2026 /PRNewswire/ — ABANZA, a company focused on next-generation soft-tissue repair solutions, today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for QuadLock™, its new knotless, bidirectional tension-adjustable fixation system for ACL reconstruction.

Outset Medical (Nasdaq: OM)+
announced today that it received FDA 510(k) clearance for its next-generation Tablo hemodialysis platform.
ALACHUA, Fla., Jan. 26, 2026 /PRNewswire/ — Amend Surgical, a medical device company focused on novel products for oral wound-care and regenerative biomaterials, today announced that it has achieved ISO 13485:2016 certification, the global standard for medical device quality management systems.

CORONADO, Calif., Jan. 26, 2026 /PRNewswire/ — Spine Innovation, LLC, a medical device startup that develops novel interbody fusion implants, announced today that is has received FDA 510(k) clearance to market the LOGIC™ Titanium Implant System.

MannKind (Nasdaq:MNKD) announced today that it received FDA approval for an update to the prescribing information for Afrezza inhaled insulin.