
Vaxxas gains Australia’s TGA licence to manufacture HD microarray patch
The patch is tailored to deliver all types of vaccines to the skin using a simple applicator.

The patch is tailored to deliver all types of vaccines to the skin using a simple applicator.

The FDA has granted Breakthrough Device Designation to Nephrodite’s Holly implantable continuous dialysis system.

TAMPERE, Finland, Dec. 14, 2025 /PRNewswire/ — Bioretec Oy (“Bioretec” or the “Company”), a pioneer in biodegradable orthopedic implants, has been granted Breakthrough Device Designation by the U.S. Food and Drug Administration (FDA) for its magnesium alloy technology-based, biodegradable RemeOs™ DrillPin.

HONG KONG, Dec. 12, 2025 /PRNewswire/ — Peijia Medical (9996.HK), a leading Chinese domestic company in the high-growth transcatheter valve therapeutics and neurovascular interventions markets, announced that its TaurusTrio Transcatheter Aortic Valve (TAV) system received approval from the National Medical Products Administration (NMPA) of China on December 11, 2025.
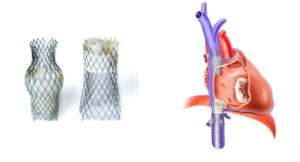
P&F USA, Inc., the U.S. subsidiary of heart valve manufacturer P&F Products and Features GmbH, today announced that the U.S. Food and Drug Administration (FDA) has approved initiation of the TRICAV II Pivotal Trial.

The Bendit17 steerable microcatheter aims to advance navigational control through complex anatomies in vascular procedures.
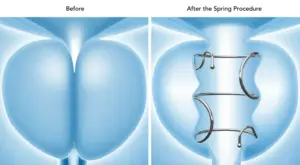
The Spring is the only FDA-approved FIT in a range of lengths and diameters, allowing for patient personalisation

PLANO, Texas, Dec. 11, 2025 /PRNewswire/ — Vesalio, a leader in thrombectomy solutions, today announced FDA 510(k) clearance and the upcoming U.S. commercial launch of enVast™, the first and only clot retriever specifically cleared for mechanical thrombectomy in the cardiac circulation. enVast introduces a proven, innovative approach to clot capture and removal, redefining coronary thrombectomy for patients with large thrombus burden (LTB).

Neurovalens announced that it received FDA de novo approval for Modius Lean, its non-invasive neurotechnology for weight management.

Flow Neuroscience’s at-home depression treatment device has become the first of its kind to be approved by the FDA.