FDA approves new stool collection kit of Geneoscopy’s CRC screening test
The test employs advanced RNA technology to identify biomarkers for colorectal cancer and advanced adenomas.

The test employs advanced RNA technology to identify biomarkers for colorectal cancer and advanced adenomas.

Simplified Process Aims to Boost Participation in Colorectal Cancer Screening and Improve Early Detection Rates

STEWARTVILLE, Minn., July 24, 2025 /PRNewswire/ — Minnesota Medical Technologies, today announced that it received FDA 510(k) clearance for its fecal incontinence device (“StaySure(™)”).
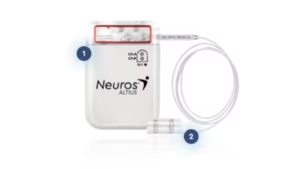
ALISO VIEJO, Calif., July 23, 2025 /PRNewswire/ — In a major milestone for chronic post-amputation pain treatment, the first commercial patient in the U.S. has been successfully implanted with the FDA-approved Altius® System – marking the beginning of the company’s mission to relieve pain and restore life for lower limb amputees.
New AI-Enabled Technology Aims to Personalize Fluid Management and Improve Outcomes Across Hospital Network
Philips (NYSE: PHG)+ announced today that it received FDA 510(k) clearance for the latest UroNav image-guided navigation system.
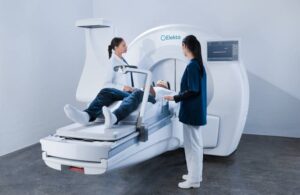
Elekta announced today that it received FDA 510(k) clearance to expand indications for its Leksell Gamma Knife radiosurgery system.
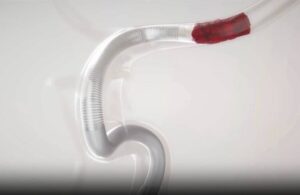
Imperative Care announced today that it received FDA 510(k) clearance for its novel Zoom 7X catheter for aspiration thrombectomy procedures.

The CE mark also allows Medtronic’s automated insulin delivery system to be used during pregnancy and by children as young as 2.

TUSTIN, Calif., July 22, 2025 /PRNewswire/ — Celluma Light Therapy, the global leader in award-winning, clinically backed LED technology, announces the launch of the Celluma NOVA, the first-of-its-kind battery-powered, shape-taking LED panel with five FDA-cleared treatment modes. The Celluma NOVA treats aging skin, pain, hair restoration, body contouring, and acne — all in a single compact, rechargeable device.