InnoCare Urologics wins FDA nod for urinary safety catheter
InnoCare Urologics announced today that it received FDA 510(k) clearance for its novel urinary safety catheter.

InnoCare Urologics announced today that it received FDA 510(k) clearance for its novel urinary safety catheter.
Beacon Biosignals announced today that it received FDA authorization for a new update to its Dreem 3S wearable sleep monitoring headband.

Nonin Medical has won FDA 510(k) clearance for its first over-the-counter fingertip pulse oximeter, the TruO2 OTC.

IRVINE, Calif., Dec. 2, 2024 /PRNewswire/ — Mentor Worldwide LLC, the number one global brand in breast aesthetics, and part of Johnson & Johnson MedTech, today announced the U.S. Food and Drug Administration (FDA) approved MENTOR™ MemoryGel™ Enhance Breast Implants for primary and revision reconstruction breast surgery in post-mastectomy women.
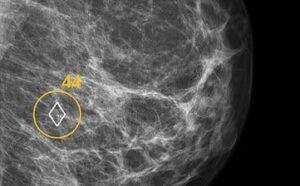
CHICAGO, Dec. 2, 2024 /PRNewswire/ — ScreenPoint Medical is showcasing a new FDA clearance for innovative new capabilities of its industry leading Breast AI Transpara here at the 110th Annual Radiological Society of North America (RSNA) meeting, December 1-4, 2024 (South Hall #5316)
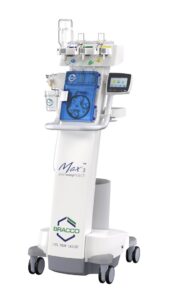
PRINCETON, N.J., Dec. 2, 2024 /PRNewswire/ — Bracco Diagnostics Inc., the U.S. subsidiary of Bracco Imaging S.p.A., a leading global company in the diagnostic imaging business, along with ulrich GmbH & Co. KG, a renowned German medical device manufacturer specializing in contrast media injectors and spinal implants, announced today the clearance of the Bracco branded Max 3™, a Rapid Exchange and Syringeless Injector for use in magnetic resonance imaging (MRI) procedures.

PLEASANTON, Calif., Dec. 2, 2024 /PRNewswire/ — Movano Health (Nasdaq: MOVE), a pioneer in health technology, today announced U.S. Food and Drug Administration (FDA) 510(k) clearance for the pulse oximeter in its EvieMED Ring.
EchoGo Amyloidosis is an AI-based software-only medical device, which aims to improve early detection of Cardiac Amyloidosis, an underdiagnosed cause of heart failure.
The device has sold well in Europe, where the company said it has a 60% market share, but will be the first product of its type available in the U.S.
SEOUL, South Korea, Nov. 25, 2024 /PRNewswire/ — Neurophet, an artificial intelligence (AI) solution company for brain disease, announced on the 25th that its brain MRI analysis software “Neurophet AQUA”, has obtained 510(k) clearance from U.S. Food and Drug Administration (FDA) for its newly integrated multiple sclerosis (MS) analysis functionality.