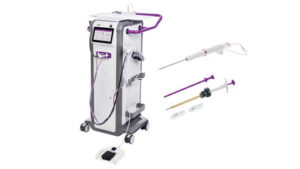
CINCINNATI, March 18, 2026 /PRNewswire/ — Mammotome, a Danaher company, announces FDA Clearance of two innovations for MR-guided breast biopsy: the Mammotome Prima™ MR Dual Vacuum-Assisted Breast Biopsy System – the industry’s first solution designed to be positioned in the MRI scanner room at the patient’s side – and the HydroMARK™ Plus Breast Biopsy Site Marker for MR, engineered for exclusive use with the Mammotome Prima™ system universal targeting set. Following a successful debut of the Mammotome Prima™ MR system in Europe, both products will be introduced to U.S. clinicians at the 2026 Society of Breast Imaging (SBI) Symposium. By bringing the device directly into the scanner room, the Mammotome Prima™ MR system enhances clinician coordination, streamlines communication, and helps maintain consistent focus on the patient throughout the procedure.