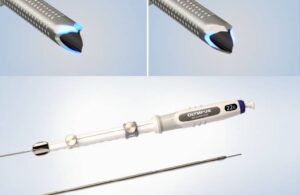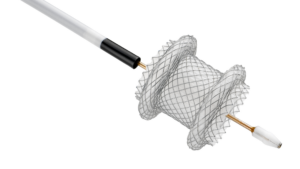
Zeus Launches PFX™ Platform and Introduces PFX Flex™ Sub-Lite-Wall™, Advancing the Future of Catheter Innovation
ORANGEBURG, S.C., Jan. 29, 2026 /PRNewswire/ — Zeus, a global leader in advanced polymer solutions, today announced the launch of PFX™, a breakthrough platform designed to advance catheter innovation with a focus on performance, design flexibility, and sustainability.