
NervGen’s Peptide Achieves Nervous System Repair in Phase II Spinal Cord Injury Study
NervGen will meet with the FDA early next year to align on a regulatory path forward for NVG-291 in chronic spinal cord injury.

NervGen will meet with the FDA early next year to align on a regulatory path forward for NVG-291 in chronic spinal cord injury.
The a2z-Unified-Triage device is designed to simultaneously flag and prioritise seven urgent findings on abdomen-pelvis CT scans.

Scientists at the University of Portsmouth have created the first detailed 3D map of how a crucial piece of connective tissue in our bodies responds to the stresses of movement and exercise. This tissue, called calcified fibrocartilage (CFC), acts like a biological shock absorber where tendons attach to bone.

Having been commercially available for less than 12 months, the system is now offered in over 60 US clinics.
AUSTIN, Texas, Nov. 18, 2025 /PRNewswire/ — Alafair Biosciences, Inc., a medical device company redefining soft-tissue protection in orthopedic surgery, announced today the first clinical use of its VersaCoat™ Flowable Hydrogel (VersaCoat™ Tendon Protector, VersaCoat™ Nerve Protector) by two leading surgeons in lower and upper extremity procedures.

Competing with companies including Stryker and J&J, Zimmer has moved to strengthen its position by developing an updated robot.
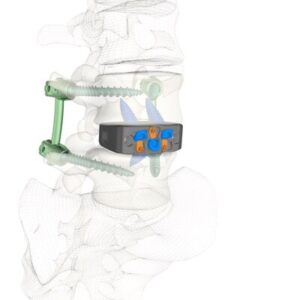
FLOWOOD, Miss., Nov. 15, 2025 /PRNewswire/ — Zavation Medical Products, LLC (“Zavation”), a leading innovator in spinal device technology, announces the first successful implantations of the Varisync® ALIF NanoPrime™ System, a next-generation stand-alone ALIF system featuring Zavation’s proprietary titanium ion bond technology.
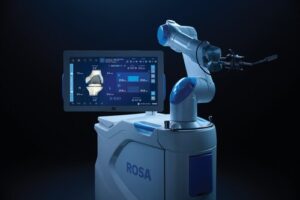
WARSAW, Ind., Nov. 14, 2025 /PRNewswire/ — Zimmer Biomet Holdings, Inc. (NYSE and SIX: ZBH), a global medical technology leader, today announced U.S. Food and Drug Administration (FDA) 510(k) clearance of ROSA® Knee with OptimiZe™, an enhanced version of its ROSA® Knee System that offers a more customized experience for surgeons to help deliver accurate and reproducible outcomes1 in robotic-assisted total knee replacement surgery.

The company will introduce the headset at the North American Spine Society Annual Meeting in the US on 14 November.

ALTSTÄTTEN, Switzerland and BOSTON, Nov. 13, 2025 /PRNewswire/ — icotec, the pioneer of implantable devices made from BlackArmor® Engineered Carbon/PEEK, today announced that the U.S. Food and Drug Administration (FDA) has cleared the CMORE® CT System for use in the cervicothoracic spine.