
Restore Robotics wins FDA nod to remanufacture new da Vinci instruments
Restore Robotics announced new FDA 510(k) clearances for the remanufacturing of two Intuitive da Vinci Xi robotic surgical instruments.

Restore Robotics announced new FDA 510(k) clearances for the remanufacturing of two Intuitive da Vinci Xi robotic surgical instruments.

Researchers at King’s College London have shown for the first time that AI can autonomously perform thrombectomy navigation in a physical lab setting, a step toward expanding access to life-saving stroke treatment. For their study, now published in IEEE Robotics and Automation Letters, the team developed a robotic system that uses AI to navigate the complex pathway through blood vessels from the leg to the brain during mechanical thrombectomy (MT).
HENDERSON, Nev., March 31, 2026 /PRNewswire/ — Restore Robotics today announced it has received additional 510(k) clearances from the U.S. Food and Drug Administration (FDA) for the remanufacturing of two more da Vinci Xi® robotic surgical instruments. This milestone expands Restore Robotics’ growing portfolio of cleared instruments and further strengthens its position as a leader in robotic instrument remanufacturing.
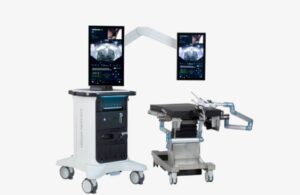
Procept BioRobotics (Nasdaq:PRCT) today announced the international rollout of its next-generation Hydros robotic system for Aquablation therapy.
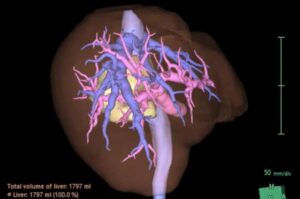
Researchers at Boston University Chobanian & Avedisian School of Medicine now show that it is possible to remove the caudate lobe safely using a surgical robot, even in an older patient, and still remove the cancer completely.
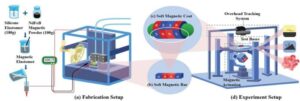
Recent technological advances have opened new possibilities for the development of advanced medical devices, including tiny robots that can safely move inside the human body. Some of these systems could help to simplify complex medical procedures, including delicate surgeries and the targeted delivery of drugs to specific sites.
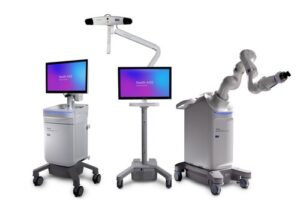
GALWAY, Ireland, Feb. 13, 2026 /PRNewswire/ — Medtronic (NYSE: MDT), a global leader in healthcare technology, today announced U.S. Food and Drug Administration (FDA) clearance of the Stealth AXiS™ surgical system, a next-generation platform that brings planning, navigation, and robotics together into a single, intelligent system for spine surgery.

Research on their new robotic crash cart (RCC)—a robotic version of the mobile drawer unit that holds supplies and equipment needed for a range of medical procedures.

Soft tissue robotic systems designed to improve surgical precision are expanding into new specialties and increasing competition for longtime industry leader Intuitive Surgical.
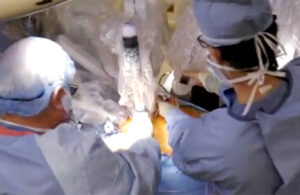
Intuitive Surgical (Nasdaq: ISRG)+
has received FDA clearance for “several cardiac procedures,” CEO Dave Rosa said today while announcing the surgical robotics company’s latest financial and operating results.