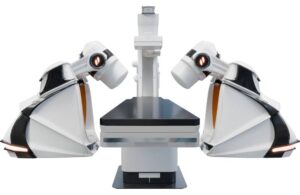
Stereotaxis wins CE mark for next-gen surgical robot, submits to FDA
Stereotaxis (NYSE:STXS) announced that it obtained CE mark in Europe and submitted an FDA 510(k) application for its next-generation surgical robot.

Stereotaxis (NYSE:STXS) announced that it obtained CE mark in Europe and submitted an FDA 510(k) application for its next-generation surgical robot.
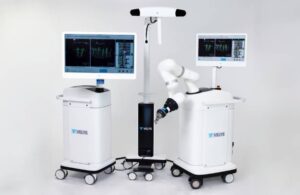
Johnson & Johnson’s (NYSE: JNJ)+
DePuy Synthes announced today that it launched a dual-use robotics and standalone navigation platform that uses its Velys robot in spine surgery.

Perceptive today announced the completion of a fully automated dental procedure on a human using its advanced robotic system.
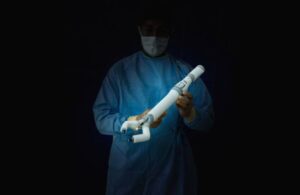
Virtual Incision today announced the completion of the first surgery in a study assessing its surgical robot for hysterectomy procedures.

A paraplegic person carried the Olympic torch as part of the traditional relay using an exoskeleton developed by Wandercraft.
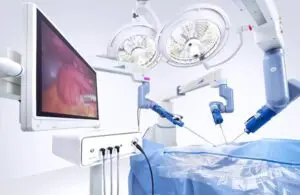
Asensus Surgical (NYSE:ASXC) announced today that it received FDA 510(k) clearance for an expanded surgical robot indication.
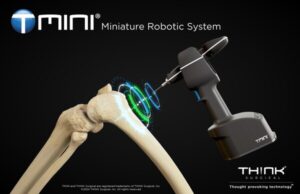
FREMONT, Calif., July 23, 2024 /PRNewswire/ — THINK Surgical, Inc., an innovator in the field of orthopedic surgical robots, today announced that its TMINI® Miniature Robotic System (TMINI 1.1) has received 510(k) clearance from the U.S. Food and Drug Administration (FDA).

eCential Robotics announced today that it received FDA 510(k) clearance for its spine navigation and robotic-assistance device.

Smith+Nephew (NYSE: SNN)+
announced today that it received FDA 510(k) clearance for its new Catalystem primary hip system.

A new surgical procedure gives people more neural feedback from their residual limb. With it, seven patients walked more naturally and navigated obstacles.