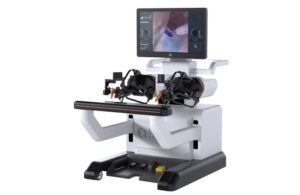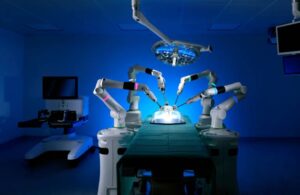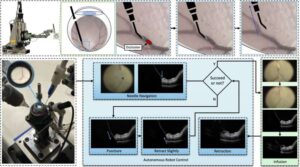
A new robotic system could perform delicate eye surgery
Retinal vein occlusion (RVO) is a severe disease that occurs when a vein in the light-sensitive layer at the back of the eye (i.e., the retina) becomes blocked, which results in a loss of vision. There are currently a few medical interventions that address RVO, including the periodic injection of medications that block the abnormal growth of blood vessels or of steroids, which reduce swelling and inflammation.