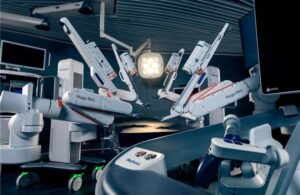
Medtronic gets FDA nod for Hugo surgical robot in urologic procedures
Medtronic (NYSE: MDT)+
announced today that the FDA cleared its Hugo robotic-assisted surgery system for use in urologic surgical procedures.

Medtronic (NYSE: MDT)+
announced today that the FDA cleared its Hugo robotic-assisted surgery system for use in urologic surgical procedures.

Medical Microinstruments (MMI) announced today that it won reimbursement for its surgical lymphovenous bypass (LVB) surgery system.
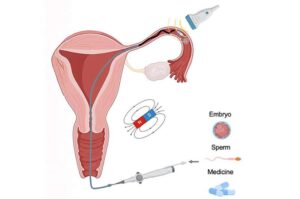
A new international study led by the Nanobiosystems group at CIC nanoGUNE, is developing miniature, non-invasive, precise robotic catheters for use in reproductive medicine and gynecological health.

Microbot is collaborating with Emory to establish an Endovascular Robotics Program in interventional radiology to enhance the growing and evolving specialty field.

What if a robot could show us how the brain keeps us balanced? UBC scientists built one—and their discovery could help shape new ways to reduce fall risk for millions of people.
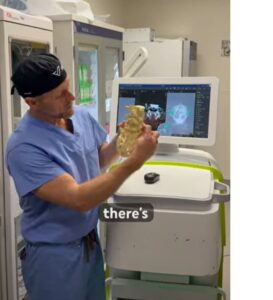
RESTON, Va., Nov. 25, 2025 /PRNewswire/ — VSI® Spine Surgeon Dr. Christopher Good has achieved a historic milestone in robotic spine surgery, performing the world’s first minimally invasive robotic Bertolotti’s resection surgery at Reston Hospital Center (HCA Virginia Health System). This first-ever procedure introduces a transformative surgical option for patients suffering from chronic low back pain caused by Bertolotti syndrome, an underdiagnosed spinal condition affecting an estimated 4-8% of the population.
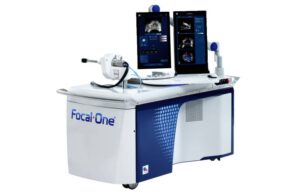
EDAP TMS SA (Nasdaq:EDAP) announced today that the FDA granted 510(k) clearance for new workflows for its ultrasound technology.
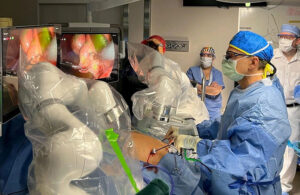
Levita Magnetics has received FDA 510(k) clearance for its Magnetic Surgical System (MSS) to be used in certain pediatric surgeries, with the first U.S. surgery performed at Cleveland Clinic Children’s earlier this month.

A decade ago, at age 55, Don Lewis suffered a stroke in his sleep. When he woke up, he couldn’t move his left arm or leg. Lewis’s neighbor realized his truck hadn’t moved in two days and called 911 for a welfare check. When paramedics found him, he was paralyzed on one side.
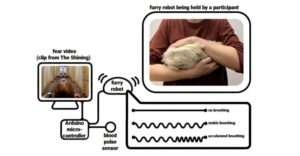
Humans can “catch” fear from robots, new research has shown. The findings—by a team of psychologists from the University of Amsterdam and the University of British Columbia—shed new light on how emotions can spread through touch, with implications for human relationships, mental health, and future technologies such as virtual reality and wearable devices.