
FDA clears Intuitive curved vessel sealer device for surgical robot
Intuitive Surgical (Nasdaq: ISRG)+
announced today that the FDA cleared the latest advanced energy instrumentation for its da Vinci systems.

Intuitive Surgical (Nasdaq: ISRG)+
announced today that the FDA cleared the latest advanced energy instrumentation for its da Vinci systems.
The FDA has granted 510(k) clearance to the TaviPilot AI software developed by Caranx Medical, according to company officials.

Mendaera has received FDA 510(k) clearance for its Focalist handheld robotic system, which enhances the precision of ultrasound-guided needle placement across multiple specialties.
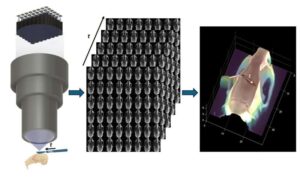
For over a century, surgeons performing delicate procedures have relied on stereoscopic microscopes to gain a sense of depth. These tools mimic human vision by presenting slightly different images to each eye, allowing the brain to perceive three-dimensional structures—a crucial aid when working with fragile blood vessels or intricate brain tissue. Despite modern upgrades like digital displays and video capture, today’s operating microscopes still depend on the same core principle: two views, interpreted by the human brain.
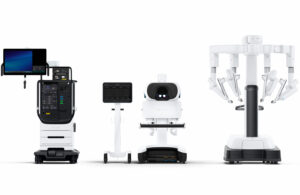
Intuitive Surgical (Nasdaq: ISRG)+
announced today that it received CE mark for its da Vinci 5 surgical robotic system.
Magnetic-Assisted Robotic Platform Now Approved for Bariatric and Hiatal Hernia Procedures, Broadening Access to Scar-Reducing Surgical Innovation

New ChoiceSpine™ App Enhances Precision and Efficiency in Spine Surgery with Op.n™ Robotic and Navigation Technology
Levita Magnetics announced today that it received expanded FDA 510(k) clearance for its MARS (magnetic-assisted robotic surgery) system.

Brain Navi Biotechnology announced today that it received FDA 510(k) clearance for its NaoTrac stereotaxic guiding surgical robotic device.
Johnson & Johnson MedTech (NYSE: JNJ)+
today announced the U.S. launch of its Ethicon 4000 surgical stapler.