
Pulse Biosciences wins FDA IDE to conduct study of nsPFA tech for AFib
Pulse Biosciences (Nasdaq:PLSE) announced today that the FDA granted investigational device exemption (IDE) for its nanosecond pulsed field ablation (nsPFA) system.

Pulse Biosciences (Nasdaq:PLSE) announced today that the FDA granted investigational device exemption (IDE) for its nanosecond pulsed field ablation (nsPFA) system.

Lifeward (Nasdaq:LFWD) announced today that it received CE mark approval for its ReWalk 7 personal exoskeleton.
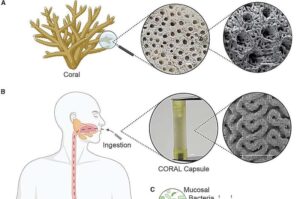
In the depths of the ocean, marine corals have evolved intricate, porous structures that shelter diverse microbial communities. Now, researchers have borrowed this biological blueprint to create an ingestible pill that can sample bacteria from one of the most inaccessible regions of the human body: the small intestine.
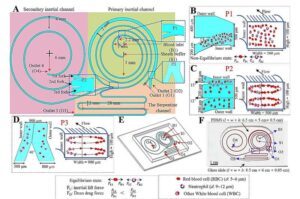
Sepsis arises from infection and immune dysregulation. Neutrophils play a key role in its progression, yet existing clinical tools cannot simultaneously isolate these cells and measure their functional activity.
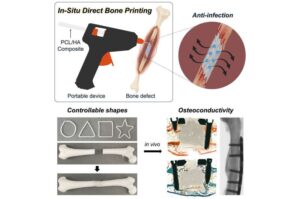
Scientists have developed a tool made from a modified glue gun that can 3D print bone grafts directly onto fractures and defects during surgery.

An ultrasound device that can precisely stimulate areas deep in the brain without surgery has been developed by researchers from UCL and the University of Oxford, opening up new possibilities for neurological research and treatment of disorders such as Parkinson’s disease.
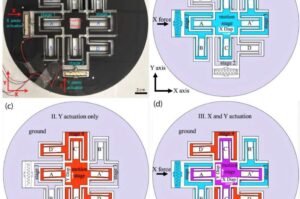
In the quest to address infertility, Cornell researchers have developed a groundbreaking device that could simplify and automate oocyte cumulus removal, a critical step in assisted reproductive technologies.
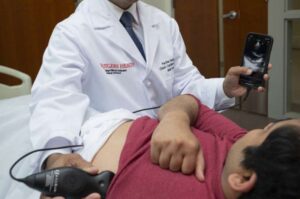
When hospitalized patients struggle to breathe, doctors typically reach for their stethoscopes, but results from a Rutgers and RWJBarnabas Health clinical study in JAMA Network Open suggest they should diagnose the problem with portable ultrasounds instead.

The company raised $250 million in July to expand its manufacturing capabilities and establish multiple teams to support the launch.

OKLAHOMA CITY, Sept. 4, 2025 /PRNewswire/ — Biozen, LLC a digital health innovator integrating advanced biosensors with proprietary algorithms, will present its cuffless blood pressure technology at the American Heart Association (AHA) Hypertension Scientific Sessions on Sept. 5 at 9:00 a.m. in Baltimore, Maryland (Poster Session 2, Poster #FR443).