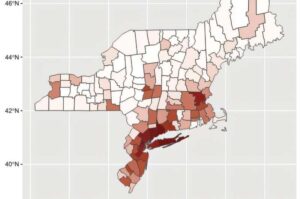
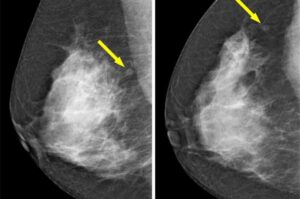
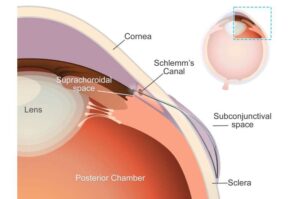
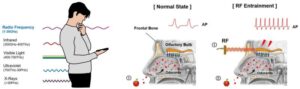
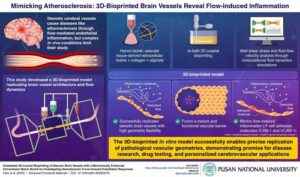
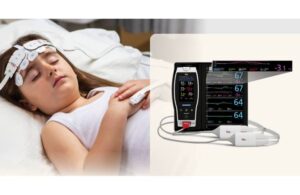
DTSS’s acoustic innovation addresses the healthcare application market projected to exceed $100 billion in China and approach $90 billion globally by 2030
Datasea, a Nevada-based technology enterprise specialising in advanced acoustic high-tech products and AI multimodal digital solutions, today announced a milestone breakthrough in its acoustic technology