
Vivani reports first human GLP-1 implant for weight loss in adults
Vivani Medical (Nasdaq:VANI) announced today that it successfully administered the first GLP-1 implant in its LIBERATE-1 clinical trial.

Vivani Medical (Nasdaq:VANI) announced today that it successfully administered the first GLP-1 implant in its LIBERATE-1 clinical trial.

Johnson & Johnson MedTech (NYSE: JNJ)+
announced today that the FDA cleared its Monarch Quest technology for robotic-assisted bronchoscopy.
Spinal cord injuries are life-altering, often leaving individuals with severe mobility impairments. While rehabilitation robotics—devices that guide movement during therapy—have improved training for those with spinal cord injuries, their effectiveness remains limited. Without active muscle engagement, robotic-assisted movement alone does not sufficiently retrain the nervous system.

SALT LAKE CITY, March 12, 2025 /PRNewswire/ — For the thousands of patients in the United States requiring treatment for bone loss or defects caused by trauma or infections, Elute, Inc., a clinical stage company and emerging leader with a groundbreaking controlled and extended drug delivery platform, proudly announces U.S. Food and Drug Administration (FDA) approval of BonVie+™, a novel bone void filler implant.
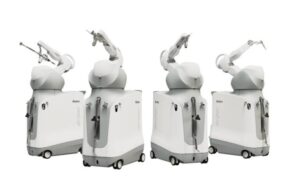
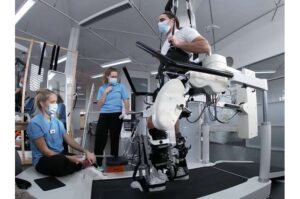
Stryker (NYSE: SYK) announced today that it is commercially launching the next generation of its Mako surgical robotic platform.

Sooma Medical announced today that it received FDA investigational device exemption (IDE) for its transcranial direct current stimulation (tDCS) device.

New research from the University of Kentucky has earned a spot on the cover of the Journal of Neuroscience, highlighting a major challenge in spinal cord injury recovery.
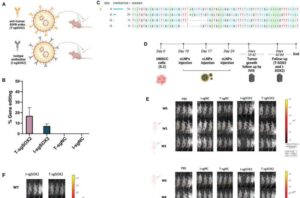
Researchers from Tel Aviv University utilized CRISPR to cut a single gene from cancer cells of head and neck tumors—and successfully eliminated 50% of the tumors in model animals. This study was led by Dr. Razan Masarwy, MD, Ph.D. from the lab of Prof. Dan Peer. The findings are published in the journal Advanced Science.
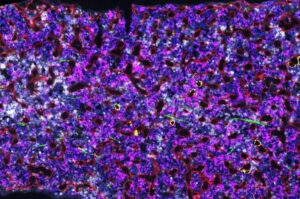
Researchers at the Francis Crick Institute have identified genetic changes in blood stem cells from frequent blood donors that support the production of new, non-cancerous cells.
Miach Orthopaedics announced today that the FDA cleared an expanded indication for the company’s Bear implant for ACL tears.