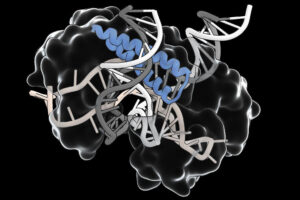
An ancient RNA-guided system could simplify delivery of gene editing therapies
The programmable proteins are compact, modular, and can be directed to modify DNA in human cells.

The programmable proteins are compact, modular, and can be directed to modify DNA in human cells.
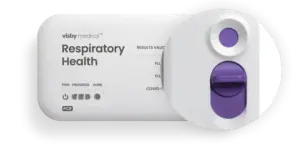
SAN JOSE, Calif., Feb. 26, 2025 /PRNewswire/ — Visby Medical™ announced today that it has received 510(k) clearance and was granted a CLIA waiver from the U.S. Food and Drug Administration (FDA) for its point-of-care respiratory health test. The Visby Medical Respiratory Health Test is a rapid polymerase chain reaction (PCR) test that detects and differentiates between upper respiratory infections caused by influenza (Flu) A & B, and SARS-CoV-2 (COVID-19). This multiplexed molecular device is the first handheld test to receive this designation after being granted Emergency Use Authorization (EUA) in December 2022.

TULSA, Okla., Feb. 26, 2025 /PRNewswire/ — Sway Medical, Inc., the company that created the Mobile Concussion Management category, is proud to announce that it has received FDA 510(k) clearance as a Computerized Cognitive Assessment Aid for Concussion under Section 882.1471. This clearance expands on Sway Medical’s previous FDA clearance for balance testing in head injuries, officially recognizing Sway as the first fully integrated tool that combines both cognitive and balance testing into one product for concussion management.

Our organs age at different rates, and a blood test determining how much they’ve each aged could predict the risk of conditions like lung cancer and heart disease decades later, finds a new study led by University College London (UCL) researchers.
ALS, or amyotrophic lateral sclerosis, can sometimes be difficult to diagnose or to predict how quickly the disease is likely to progress. A new study helps determine which blood tests are best at identifying and monitoring ALS. The study is published in the online issue of Neurology.
UCLA scientists have identified a potential new strategy for treating glioblastoma, the deadliest form of brain cancer, by reprogramming aggressive cancer cells into harmless ones.

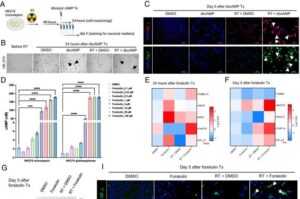
When scientists stimulated cells to produce a protein that helps “water bears” survive extreme environments, the tissue showed much less DNA damage after radiation treatment.

PALM COAST, Fla., Feb. 25, 2025 /PRNewswire/ — OrthoNovis, Inc is proud to announce that it has received U.S. Food and Drug Administration (FDA) clearance to market its innovative BPS Wrist Fracture System. www.orthonovis.com
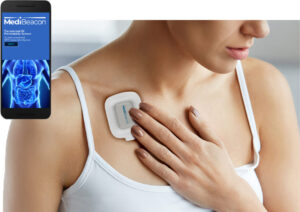
ST. LOUIS, Feb. 25, 2025 /PRNewswire/ — MediBeacon Inc. today announced the National Medical Products Administration (NMPA) in China has approved the MediBeacon® TGFR Monitor and TGFR Sensor for the assessment of kidney function in patients with normal or impaired renal function. Lumitrace® (relmapirazin) injection, categorized as a drug in China, is under review and is targeted for approval in late 2025. The transdermal GFR technology includes Lumitrace (relmapirazin) injection, a non-radioactive, non-iodinated fluorescent GFR tracer agent, which together with the TGFR Monitor and TGFR Sensor allow assessment of kidney function by measuring the clearance rate of the fluorescent agent as it leaves the body.
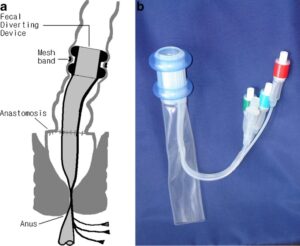
SAN FRANCISCO, Feb. 25, 2025 /PRNewswire/ — Averto Medical, a clinical-stage medical device company pioneering minimally invasive gastrointestinal care, is pleased to announce that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation for its ColoSeal™ Intraluminal Colonic Diversion (ICD) System. This designation underscores the potential of ColoSeal™ to significantly improve outcomes for patients undergoing colorectal surgery by eliminating the need for a temporary ostomy.