
FDA clears AI 3D head, neck imaging tech from RapidAI
RapidAI announced that it received FDA 510(k) clearance for its Lumina 3D automated 3D imaging reconstruction solution.

RapidAI announced that it received FDA 510(k) clearance for its Lumina 3D automated 3D imaging reconstruction solution.

Technology is available to assist with nearly every facet of modern life, including helping to prevent people with dementia from wandering and alerting their caregivers when they do.

A research group from the Institute for Research on Next-generation Semiconductor and Sensing Science (IRES²) at Toyohashi University of Technology has developed an innovative in vivo electrophysiological neural recording technology that minimizes neuronal death and allows stable recordings for over a year.
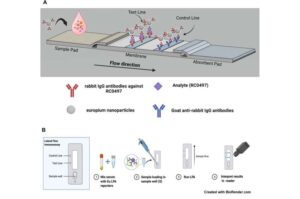
A team of biomolecular engineers, pathologists, and internal medicine specialists at the University of Texas Medical Branch, working with a colleague from the University of Houston, has developed a quick test for tick-borne spotted fever rickettsioses (SFRs).

INDIANAPOLIS, Jan. 29, 2025 /PRNewswire/ — Roche announced today that the Tina-quant® Lipoprotein (a) Gen.2 Molarity assay has received 510(k) clearance from the United States Food and Drug Administration (FDA). This will be the first 510(k) cleared test of its kind available in the U.S. measuring lipoprotein (a), or Lp(a), in nanomoles per liter (nmol/L). The National Lipid Association (NLA) recommends all adults measure their Lp(a) – often referred to as L-P-Little-A – at least once in a lifetime to help assess cardiovascular risk.
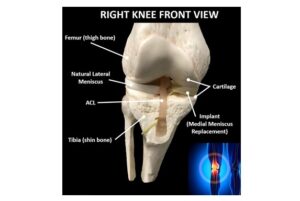
RICHMOND, Va. and ATLANTA, Jan. 29, 2025 /PRNewswire/ — OrthoPreserve, a company developing orthopedic implant solutions, announced today it has been granted both a Breakthrough Device Designation and Total Product Life Cycle Advisory Program (TAP) enrollment from the U.S. Food & Drug Administration (FDA) for Defender, a meniscus replacement implant.
Researchers have developed a powerful tool that can detect variants of SARS-CoV-2 with high transmission potential before they become widespread. This approach could significantly support public health efforts to control outbreaks and help identify new variants that need closer monitoring.
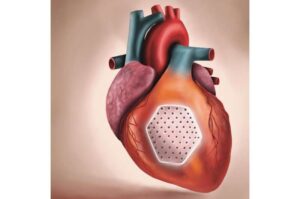
The heart patch is produced from induced pluripotent stem cells derived from heart muscle cell.
NEW YORK, Jan. 28, 2025 /PRNewswire/ — AI Optics Inc., an AI-focused medical device company announced today that it has received FDA 510(k) clearance for its Sentinel Camera. This handheld retinal imaging system captures high-quality images of the human eye, helping to address critical gaps in retinal disease screening through a portable and accessible solution.
BREA, Calif., Jan. 28, 2025 /PRNewswire/ — Beckman Coulter Diagnostics, a global leader in clinical diagnostics, today announced the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation to Beckman Coulter’s Access p‑Tau217/β-Amyloid 1-42 plasma ratio. This blood test is designed to aid healthcare providers identify patients with amyloid pathology associated with Alzheimer’s disease.