
CergenX wins FDA breakthrough nod for neonatal brain monitoring tech
CergenX announced today that the FDA granted breakthrough device designation for its Wave device for neonatal brain monitoring.

CergenX announced today that the FDA granted breakthrough device designation for its Wave device for neonatal brain monitoring.

MedRhythms announced that it received a final pricing determination from CMS for its InTandem rehabilitation system.

AliveCor announced that CMS included its AI-powered ECG system in a final rule that sets a Medicare payment rate for the technology.

Micron Biomedical announced today that it added $16 million in funding to bring its Series A equity round to more than $33 million.

GT Medical Technologies announced today that it completed a $37 million first close of a Series D financing round.

CardioFocus today announced the publication of 12-month data from a study of its pulsed field ablation (PFA) system.
Medtronic (NYSE: MDT)+
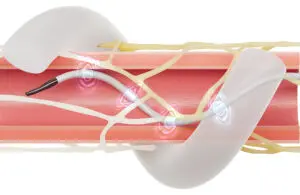
announced that CMS is opening a national coverage analysis on renal denervation (RDN) procedures for patients with hypertension.
An Abbott (NYSE: ABT)+
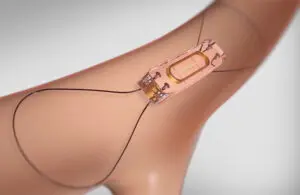
official said on social media that the company secured a significant Medicare win for its CardioMEMS platform.

Biobot Surgical announced today that it received CE mark for its Mona Lisa 2.0 surgical robotic platform for urology applications.

TEL AVIV, Israel, Jan. 14, 2025 /PRNewswire/ — BAIBYS™, a pioneering Israeli startup in AI-powered micro-robotics for Artificial Reproductive Technology (ART), today announced it has received the CE mark for its innovative BAIBYS™ system.