
FDA approves Insightec Exablate Prime for use on Philips systems
Insightec announced today that it received FDA approval and CE mark for its Exablate Prime system with certain Philips MR systems.

Insightec announced today that it received FDA approval and CE mark for its Exablate Prime system with certain Philips MR systems.

Route 92 Medical announced today that it received CE mark for its line of neurovascular intervention products for treating stroke.

The RESPOND ventilator expands access to care with a 5-year warranty and minimal maintenance needs, enhancing long-term affordability.

President-elect Donald Trump today said he is nominating Dr. Mehmet Oz to serve as administrator of the Centers for Medicare & Medicaid Services (CMS).
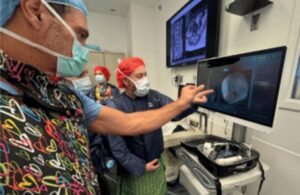
Augmedics announced today that the FDA cleared the CT-to-fluoroscopy (CT-Fluoro) registration method for its Xvision Spine System.
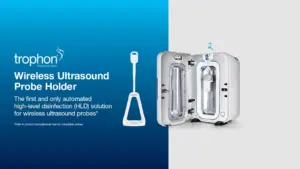
SYDNEY, Nov. 19, 2024 /PRNewswire/ — Nanosonics, Inc., a global leader in infection prevention solutions, is proud to announce the release of its newest accessory for the US and Canadian markets—the trophon® Wireless Ultrasound Probe Holder.

FLOWER MOUND, Texas, USA, Nov. 19, 2024 /PRNewswire/ — Stryker (NYSE: SYK), a global leader in medical technologies, unveiled the Oculan Lighting Platform, an innovative lighting solution designed to provide consistent, high-quality illumination, allowing surgeons to focus on delivering the highest standard of care.
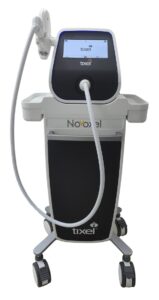
Simple Relief™ from Dry Eye comes from 2-minute treatment with Tixel i®

Zimmer Biomet (NYSE:ZBH) announced today that it received CE mark approval in Europe for its Persona Revision Knee system.

FREMONT, Calif., Nov. 18, 2024 /PRNewswire/ — THINK Surgical, Inc., an innovator in the field of orthopedic surgical robots, today announced that its TMINI® Miniature Robotic System has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for use with the LinkSymphoKnee (LSK) from Waldemar Link GmbH & Co. KG, Germany (LINK) under a Collaboration Agreement between the two companies.