
GE HealthCare’s MIM Software wins FDA clearance for Monte Carlo dosimetry
GE HealthCare’s MIM Software today announced it received FDA 510(k) clearance for Monte Carlo dosimetry.

GE HealthCare’s MIM Software today announced it received FDA 510(k) clearance for Monte Carlo dosimetry.
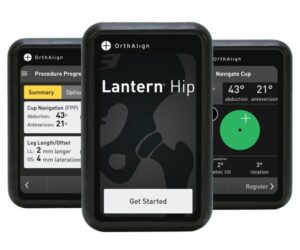
IRVINE, Calif., Oct. 3, 2024 /PRNewswire/ — OrthAlign, Inc., a privately held medical device company, announces FDA 510(k) clearance of their Lantern Hip handheld technology for direct anterior total hip arthroplasty with the patient in the supine position. Lantern Hip is the latest addition to the Lantern platform, joining existing applications for total knee, revision knee, and partial knee arthroplasty.

A 2025 commercial launch is planned to expand access to this platform technology.
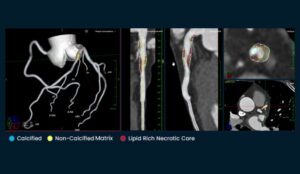
The company says this is the first and only software validated on ground-truth histology, the gold standard for plaque characterisation according to the company.

The tiny device uses a tightly focused beam of light to capture and manipulate cells.

Eyenovia (Nasdaq:EYEN) announced that it began manufacturing batches of its Mydcombi product for its next-generation delivery platform.
SetPoint Medical today announced FDA Investigational Device Exemption approval to study its proprietary neuroimmune modulation platform for people with relapsing-remitting multiple sclerosis.
AtriCure (Nasdaq: ATRC)+
announced that it received CE mark for the EnCompass clamp for cardiac tissue ablation procedures.
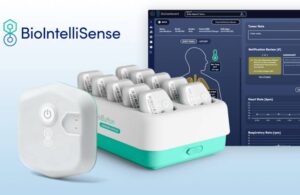
BioIntelliSense announced today that it received FDA clearance for its rechargeable BioButton multi-patient wearable and BioDashboard system.
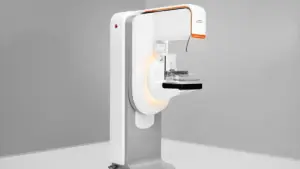
The 3D capabilities are part of Siemens’ first completely redesigned mammography platform in over a decade.