
Pulse Biosciences touts first-in-human pulsed field ablation cases
Pulse Biosciences (Nasdaq:PLSE) today announced the first completed cases in a first-in-human feasibility study of its pulsed field ablation system.

Pulse Biosciences (Nasdaq:PLSE) today announced the first completed cases in a first-in-human feasibility study of its pulsed field ablation system.
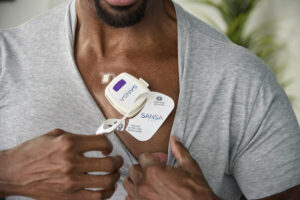
ATLANTA, Aug. 7, 2024 /PRNewswire/ — Huxley Medical, a developer of technologies that streamline cardiopulmonary care, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its groundbreaking, chest-worn, sleep apnea diagnostic patch, SANSA.

MIDDLETON, Wis., Aug. 7, 2024 /PRNewswire/ — Natus Medical Incorporated has announced the launch of autoSCORE, the first-of-its-kind artificial intelligence model capable of automatic and comprehensive clinical EEG interpretation, providing accuracy on par with medical experts.
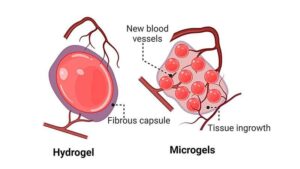
UNSW researchers have developed a new microgel made of moth silk that is designed to assist with tissue regeneration and could help people recover from heart attacks.

Researchers have developed an inexpensive bandage that uses an electric field to promote healing in chronic wounds

Researchers at Stanford Engineering have developed a nanoparticle platform that could make existing vaccines more effective, including those for influenza, COVID-19, and HIV.
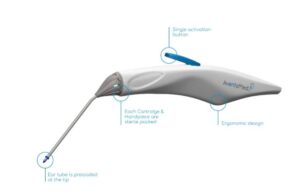
AventaMed, a Karl Storz company, announced that it received FDA 510(k) clearance for its Solo+ ear tube placement system.

Medtronic (NYSE: MDT)+ got another boost for its CGM portfolio, adding to the collaboration with Abbott announced today.
CINCINNATI, OH August 7, 2024 – Ethicon*, a Johnson & Johnson MedTech company**, announced today that the U.S. Food & Drug Administration (FDA) has approved a label update to expand the availability of the LINX™ Reflux Management System to include patients with Barrett’s esophagus (BE) experiencing gastroesophageal reflux disease (GERD) symptoms.

IRVINE, Calif., Aug. 6, 2024 /PRNewswire/ — Aspen Medical Products (Aspen), the industry leader in spine solutions for pain and mobility management, has launched the Align Orthosis. In collaboration with spine surgeon Han Jo Kim, MD of the Hospital for Special Surgery, the all-new Align Orthosis is the first prefabricated TLSO designed to help mitigate post-surgical incidence of proximal junctional kyphosis (PJK).