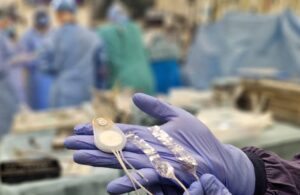
CorTec’s brain-computer interface (BCI) has obtained breakthrough device designation from the US Food and Drug Administration (FDA) for use in stroke rehabilitation.
Under development for brain-based communication and neural recording, the breakthrough designation covers the Germany company’s implantable BCI for the provision of direct cortical electrical stimulation, on the basis of recorded brain signals, to support motor recovery in people living with chronic stroke-related impairments.
According to CorTec, it is the first BCI developer to be granted an FDA breakthrough designation for such an indication.